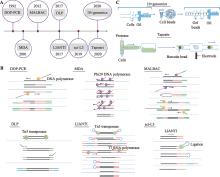
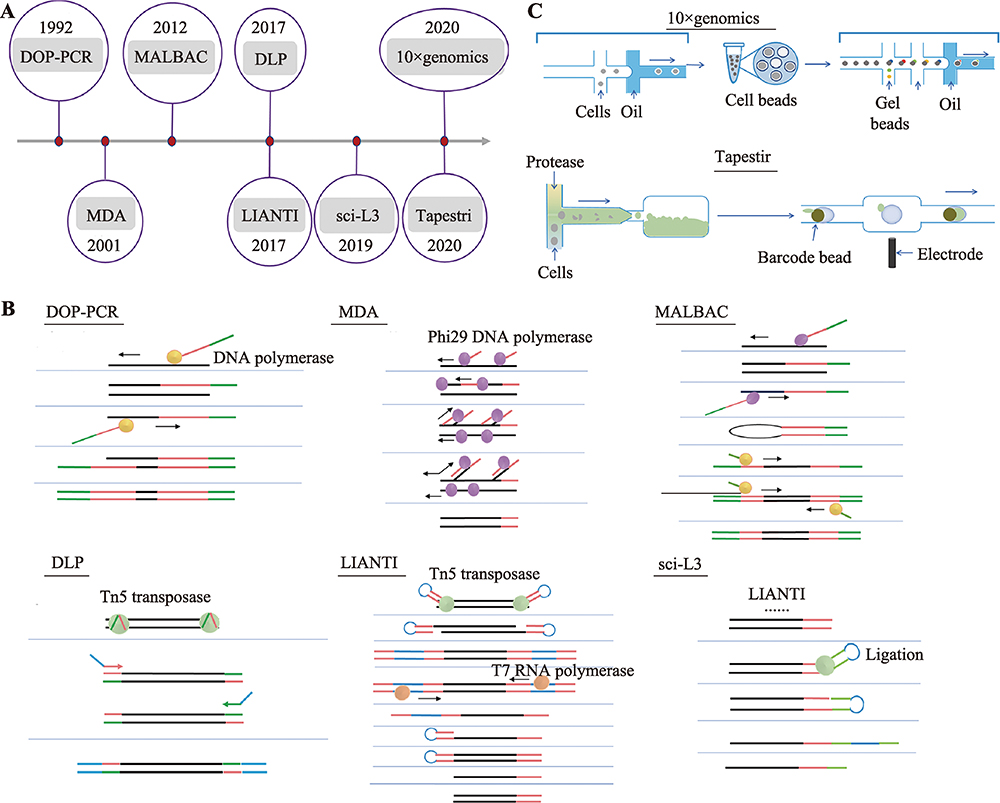
Hereditas(Beijing) ›› 2021, Vol. 43 ›› Issue (2): 108-117.doi: 10.16288/j.yczz.20-363
• Review • Previous Articles Next Articles
Advances in single-cell whole genome sequencing technology and its application in biomedicine
Zhuo Wang1( ), Xiaohan Shen1, Qihui Shi1(
), Xiaohan Shen1, Qihui Shi1( )
)
- 1 Institutes of Biomedical Sciences, Fudan University, Shanghai 201100, China
-
Received:2020-12-01Online:2021-02-16Published:2021-01-22 -
Supported by:China Postdoctoral Science Foundation Grant(2019M651377);Shanghai “Super Postdoctoral Fellow” Program(2018-2020)
Cite this article
Zhuo Wang, Xiaohan Shen, Qihui Shi.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] | Zhou Y, Wang CJ, Zhu CC, Chen JR, Cheng M, Deng YL, Guo Y. Single-cell gene variation analysis method for single gland. Hereditas(Beijing) , 2017, 39(8): 753-762. |
| 周彦, 王超杰, 朱纯超, 陈江荣, 程酩, 邓宇亮, 郭妍. 组织单腺体内单细胞的基因变异分析方法. 遗传, 2017, 39(8): 753-762. | |
| [2] | Zhao LN, Wang N, Yang GL, Su XB, Han ZG. A method for reliable detection of genomic point mutations based on single-cell target-sequencing. Hereditas(Beijing) , 2020, 42(7): 703-712. |
| 赵利楠, 王娜, 杨国良, 苏现斌, 韩泽广. 基于单细胞靶向测序探究基因碱基突变的方法. 遗传, 2020, 42(7): 703-712. | |
| [3] | Nagano T, Lubling Y, Stevens TJ, Schoenfelder S, Yaffe E, Dean W, Laue ED, Tanay A, Fraser P. Single-cell Hi-C reveals cell-to-cell variability in chromosome structure. Nature , 2013, 502(7469): 59-64. |
| [4] |
Babbe H, Roers A, Waisman A, Lassmann H, Goebels N, Hohlfeld R, Friese M, Schröder R, Deckert M, Schmidt S, Ravid R, Rajewsky K. Clonal expansions of CD8(+) T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. J Exp Med , 2000, 192(3): 393-404.
doi: 10.1084/jem.192.3.393 pmid: 10934227 |
| [5] | Dalerba P, Kalisky T, Sahoo D, Rajendran PS, Rothenberg ME, Leyrat AA, Sim S, Okamoto J, Johnston DM, Qian DL, Zabala M, Bueno J, Neff NF, Wang JB, Shelton AA, Visser B, Hisamori S, Shimono Y, van de Wetering M, Clevers H, Clarke MF, Quake SR. Single-cell dissection of transcriptional heterogeneity in human colon tumors. Nat Biotechnol , 2011, 29(12): 1120-1127. |
| [6] | Frumkin D, Wasserstrom A, Itzkovitz S, Harmelin A, Rechavi G, Shapiro E. Amplification of multiple genomic loci from single cells isolated by laser micro-dissection of tissues. BMC Biotechnol , 2008, 8: 17. |
| [7] | Bhattacherjee V, Mukhopadhyay P, Singh S, Roberts EA, Hackmiller RC, Greene RM, Pisano MM. Laser capture microdissection of fluorescently labeled embryonic cranial neural crest cells. Genesis , 2004, 39(1): 58-64. |
| [8] |
Marcus JS, Anderson WF, Quake SR. Microfluidic single-cell mRNA isolation and analysis. Anal Chem , 2006, 78(9): 3084-3089.
pmid: 16642997 |
| [9] | White AK, VanInsberghe M, Petriv OI, Hamidi M, Sikorski D, Marra MA, Piret J, Aparicio S, Hansen CL. High-throughput microfluidic single-cell RT-qPCR. Proc Natl Acad Sci USA , 2011, 108(34): 13999-14004. |
| [10] |
Pushkarev D, Neff NF, Quake SR. Single-molecule sequencing of an individual human genome. Nat Biotechnol , 2009, 27(9): 847-850.
doi: 10.1038/nbt.1561 pmid: 19668243 |
| [11] | Snabes MC, Chong SS, Subramanian SB, Kristjansson K, DiSepio D, Hughes MR. Preimplantation single-cell analysis of multiple genetic loci by whole-genome amplification. Proc Natl Acad Sci USA , 1994, 91(13): 6181-6185. |
| [12] | Zhang L, Cui XF, Schmitt K, Hubert R, Navidi W, Arnheim N. Whole genome amplification from a single cell: implications for genetic analysis. Proc Natl Acad Sci USA , 1992, 89(13): 5847-5851. |
| [13] | Telenius H, Carter NP, Bebb CE, Nordenskjöld M, Ponder BA, Tunnacliffe A. Degenerate oligonucleotide-primed PCR: general amplification of target DNA by a single degenerate primer. Genomics , 1992, 13(2): 718-725. |
| [14] | Cheung VG, Nelson SF. Whole genome amplification using a degenerate oligonucleotide primer allows hundreds of genotypes to be performed on less than one nanogram of genomic DNA. Proc Natl Acad Sci USA , 1996, 93(25): 14676-14679. |
| [15] | Klein CA, Schmidt-Kittler O, Schardt JA, Pantel K, Speicher MR, Riethmüller G. Comparative genomic hybridization, loss of heterozygosity, and DNA sequence analysis of single cells. Proc Natl Acad Sci USA , 1999, 96(8): 4494-4499. |
| [16] |
Navin N, Kendall J, Troge J, Andrews P, Rodgers L, McIndoo J, Cook K, Stepansky A, Levy D, Esposito D, Muthuswamy L, Krasnitz A, McCombie WR, Hicks J, Wigler M. Tumour evolution inferred by single-cell sequencing. Nature , 2011, 472(7341): 90-94.
doi: 10.1038/nature09807 pmid: 21399628 |
| [17] | Dean FB, Nelson JR, Giesler TL, Lasken RS. Rapid amplification of plasmid and phage DNA using Phi 29 DNA polymerase and multiply-primed rolling circle amplification. Genome Res , 2001, 11(6): 1095-1099. |
| [18] | Dean FB, Hosono S, Fang LH, Wu XH, Faruqi AF, Bray-Ward P, Sun ZY, Zong QL, Du YF, Du J, Driscoll M, Song WM, Kingsmore SF, Egholm M, Lasken RS. Comprehensive human genome amplification using multiple displacement amplification. Proc Natl Acad Sci USA , 2002, 99(8): 5261-5266. |
| [19] |
de Bourcy CF, De Vlaminck I, Kanbar JN, Wang JB, Gawad C, Quake SR. A quantitative comparison of single-cell whole genome amplification methods. PLoS One , 2014, 9(8): e105585.
doi: 10.1371/journal.pone.0105585 pmid: 25136831 |
| [20] | Lasken RS, Stockwell TB. Mechanism of chimera formation during the multiple displacement amplification reaction. BMC Biotechnol , 2007, 7: 19. |
| [21] | Fu YS, Li CM, Lu SJ, Zhou WX, Tang FC, Xie XS, Huang YY. Uniform and accurate single-cell sequencing based on emulsion whole-genome amplification. Proc Natl Acad Sci USA , 2015, 112(38): 11923-11928. |
| [22] | Zong CH, Lu SJ, Chapman AR, Xie XS. Genome-wide detection of single-nucleotide and copy-number variations of a single human cell. Science , 2012, 338(6114): 1622-1626. |
| [23] | Yao YX, La YF, Di R, Liu QY, Hu WP, Wang XY, Chu MX. Comparison of different single cell whole genome amplification methods and MALBAC applications in assisted reproduction. Hereditas(Beijing) , 2018, 40(8): 620-631. |
| 姚雅馨, 喇永富, 狄冉, 刘秋月, 胡文萍, 王翔宇, 储明星. 不同单细胞全基因组扩增方法的比较及MALBAC在辅助生殖中的应用. 遗传, 2018, 40(8): 620-631. | |
| [24] |
Picelli S, Björklund AK, Reinius B, Sagasser S, Winberg G, Sandberg R. Tn5 transposase and tagmentation procedures for massively scaled sequencing projects. Genome Res , 2014, 24(12): 2033-2040.
doi: 10.1101/gr.177881.114 |
| [25] |
Chen CY, Xing D, Tan LZ, Li H, Zhou GY, Huang L, Xie XS. Single-cell whole-genome analyses by Linear Amplification via Transposon Insertion (LIANTI). Science , 2017, 356(6334): 189-194.
pmid: 28408603 |
| [26] |
Zahn H, Steif A, Laks E, Eirew P, VanInsberghe M, Shah SP, Aparicio S, Hansen CL. Scalable whole-genome single-cell library preparation without preamplification. Nat Methods , 2017, 14(2): 167-173.
pmid: 28068316 |
| [27] | Vitak SA, Torkenczy KA, Rosenkrantz JL, Fields AJ, Christiansen L, Wong MH, Carbone L, Steemers FJ, Adey A. Sequencing thousands of single-cell genomes with combinatorial indexing. Nat Methods , 2017, 14(3): 302-308. |
| [28] |
Yin Y, Jiang Y, Lam KWG, Berletch JB, Disteche CM, Noble WS, Steemers FJ, Camerini-Otero RD, Adey AC, Shendure J. High-throughput single-cell sequencing with linear amplification. Mol Cell , 2019, 76(4): 676-690.e10.
pmid: 31495564 |
| [29] |
Pellegrino M, Sciambi A, Treusch S, Durruthy-Durruthy R, Gokhale K, Jacob J, Chen TX, Geis JA, Oldham W, Matthews J, Kantarjian H, Futreal PA, Patel K, Jones KW, Takahashi K, Eastburn DJ. High-throughput single-cell DNA sequencing of acute myeloid leukemia tumors with droplet microfluidics. Genome Res , 2018, 28(9): 1345-1352.
pmid: 30087104 |
| [30] |
Velazquez-Villarreal EI, Maheshwari S, Sorenson J, Fiddes IT, Kumar V, Yin YF, Webb MG, Catalanotti C, Grigorova M, Edwards PA, Carpten JD, Craig DW. Single-cell sequencing of genomic DNA resolves sub-clonal heterogeneity in a melanoma cell line. Commun Biol , 2020, 3(1): 318.
pmid: 32587328 |
| [31] | Klein CA, Schmidt-Kittler O, Schardt JA, Pantel K, Speicher MR, Riethmüller G. Comparative genomic hybridization, loss of heterozygosity, and DNA sequence analysis of single cells. Proc Natl Acad Sci USA , 1999, 96(8): 4494-4499. |
| [32] |
Martelotto LG, Baslan T, Kendall J, Geyer FC, Burke KA, Spraggon L, Piscuoglio S, Chadalavada K, Nanjangud G, Ng CKY, Moody P, D'Italia S, Rodgers L, Cox H, da Cruz Paula A, Stepansky A, Schizas M, Wen HY, King TA, Norton L, Weigelt B, Hicks JB, Reis-Filho JS. Whole-genome single-cell copy number profiling from formalin-fixed paraffin-embedded samples. Nat Med , 2017, 23(3): 376-385.
doi: 10.1038/nm.4279 pmid: 28165479 |
| [33] |
Gao RL, Davis A, McDonald TO, Sei E, Shi XQ, Wang Y, Tsai PC, Casasent A, Waters J, Zhang H, Meric-Bernstam F, Michor F, Navin NE. Punctuated copy number evolution and clonal stasis in triple-negative breast cancer. Nat Genet , 2016, 48(10): 1119-1130.
doi: 10.1038/ng.3641 pmid: 27526321 |
| [34] | Casasent AK, Schalck A, Gao RL, Sei E, Long A, Pangburn W, Casasent T, Meric-Bernstam F, Edgerton ME, Navin NE. Multiclonal invasion in breast tumors identified by topographic single cell sequencing. Cell , 2018, 172( 1-2): 205-217.e212. |
| [35] |
Xu X, Hou Y, Yin XY, Bao L, Tang AF, Song LT, Li FQ, Tsang S, Wu K, Wu HJ, He WM, Zeng L, Xing MJ, Wu RH, Jiang H, Liu X, Cao DD, Guo GW, Hu XD, Gui YT, Li ZS, Xie WY, Sun XJ, Shi M, Cai ZM, Wang B, Zhong MM, Li JX, Lu ZH, Gu N, Zhang XQ, Goodman L, Bolund L, Wang J, Yang HM, Kristiansen K, Dean M, Li YR, Wang J. Single-cell exome sequencing reveals single- nucleotide mutation characteristics of a kidney tumor. Cell , 2012, 148(5): 886-895.
doi: 10.1016/j.cell.2012.02.025 |
| [36] |
Hou Y, Song LT, Zhu P, Zhang B, Tao Y, Xu X, Li FQ, Wu K, Liang J, Shao D, Wu HJ, Ye XF, Ye C, Wu RH, Jian M, Chen Y, Xie W, Zhang RR, Chen L, Liu X, Yao XT, Zheng HC, Yu C, Li QB, Gong ZL, Mao M, Yang X, Yang L, Li JX, Wang W, Lu ZH, Gu N, Laurie G, Bolund L, Kristiansen K, Wang J, Yang HM, Li Y, Zhang XQ, Wang J. Single-cell exome sequencing and monoclonal evolution of a JAK2-negative myeloproliferative neoplasm. Cell , 2012, 148(5): 873-885.
doi: 10.1016/j.cell.2012.02.028 |
| [37] |
Wang Y, Waters J, Leung ML, Unruh A, Roh W, Shi XQ, Chen K, Scheet P, Vattathil S, Liang H, Multani A, Zhang H, Zhao R, Michor F, Meric-Bernstam F, Navin NE. Clonal evolution in breast cancer revealed by single nucleus genome sequencing. Nature , 2014, 512(7513): 155-160. Sequencing studies of breast tumour cohorts have identified many prevalent mutations, but provide limited insight into the genomic diversity within tumours. Here we developed a whole-genome and exome single cell sequencing approach called nuc-seq that uses G2/M nuclei to achieve 91% mean coverage breadth. We applied this method to sequence single normal and tumour nuclei from an oestrogen-receptor-positive(ER+) breast cancer and a triple-negative ductal carcinoma. In parallel, we performed single nuclei copy number profiling. Our data show that aneuploid rearrangements occurred early in tumour evolution and remained highly stable as the tumour masses clonally expanded. In contrast, point mutations evolved gradually, generating extensive clonal diversity. Using targeted single-molecule sequencing, many of the diverse mutations were shown to occur at low frequencies (<10%) in the tumour mass. Using mathematical modelling we found that the triple-negative tumour cells had an increased mutation rate (13.3x), whereas the ER+ tumour cells did not. These findings have important implications for the diagnosis, therapeutic treatment and evolution of chemoresistance in breast cancer.
doi: 10.1038/nature13600 |
| [38] | McMahon CM, Ferng T, Canaani J, Wang ES, Morrissette JJD, Eastburn DJ, Pellegrino M, Durruthy-Durruthy R, Watt CD, Asthana S, Lasater EA, DeFilippis R, Peretz CAC, McGary LHF, Deihimi S, Logan AC, Luger SM, Shah NP, Carroll M, Smith CC, Perl AE. Clonal selection with Ras pathway activation mediates secondary clinical resistance to selective FLT3 inhibition in acute myeloid leukemia. Cancer Discov , 2019, 9(8): 1050-1063. |
| [39] |
Demeulemeester J, Kumar P, Møller EK, Nord S, Wedge DC, Peterson A, Mathiesen RR, Fjelldal R, Zamani Esteki M, Theunis K, Fernandez Gallardo E, Grundstad AJ, Borgen E, Baumbusch LO, Børresen-Dale AL, White KP, Kristensen VN, Van Loo P, Voet T, Naume B. Tracing the origin of disseminated tumor cells in breast cancer using single-cell sequencing. Genome Biol , 2016, 17(1): 250.
doi: 10.1186/s13059-016-1109-7 pmid: 27931250 |
| [40] | Riebensahm C, Joosse SA, Mohme M, Hanssen A, Matschke J, Goy Y, Witzel I, Lamszus K, Kropidlowski J, Petersen C, Kolb-Kokocinski A, Sauer S, Borgmann K, Glatzel M, Müller V, Westphal M, Riethdorf S, Pantel K, Wikman H. Clonality of circulating tumor cells in breast cancer brain metastasis patients. Breast Cancer Res , 2019, 21(1): 101. |
| [41] | Chemi F, Rothwell DG, McGranahan N, Gulati S, Abbosh C, Pearce SP, Zhou C, Wilson GA, Jamal-Hanjani M, Birkbak N, Pierce J, Kim CS, Ferdous S, Burt DJ, Slane-Tan D, Gomes F, Moore D, Shah R, Al Bakir MA, Hiley C, Veeriah S, Summers Y, Crosbie P, Ward S, Mesquita B, Dynowski M, Biswas D, Tugwood J, Blackhall F, Miller C, Hackshaw A, Brady G, Swanton C, Dive C, Consortium T. Pulmonary venous circulating tumor cell dissemination before tumor resection and disease relapse. Nat Med , 2019, 25(10): 1534-1539. |
| [42] | Ni XH, Zhuo ML, Su Z, Duan JC, Gao Y, Wang ZJ, Zong CH, Bai H, Chapman AR, Zhao J, Xu LY, An TT, Ma Q, Wang YY, Wu MN, Sun Y, Wang SH, Li ZX, Yang XD, Yong J, Su XD, Lu YY, Bai F, Xie XS, Wang J. Reproducible copy number variation patterns among single circulating tumor cells of lung cancer patients. Proc Natl Acad Sci USA , 2013, 110(52): 21083-21088. |
| [43] |
Carter L, Rothwell DG, Mesquita B, Smowton C, Leong HS, Fernandez-Gutierrez F, Li YY, Burt DJ, Antonello J, Morrow CJ, Hodgkinson CL, Morris K, Priest L, Carter M, Miller C, Hughes A, Blackhall F, Dive C, Brady G. Molecular analysis of circulating tumor cells identifies distinct copy-number profiles in patients with chemosensitive and chemorefractory small-cell lung cancer. Nat Med , 2017, 23(1): 114-119.
doi: 10.1038/nm.4239 pmid: 27869802 |
| [44] |
Su Z, Wang ZJ, Ni XH, Duan JC, Gao Y, Zhuo ML, Li RY, Zhao J, Ma Q, Bai H, Chen HY, Wang SH, Chen XX, An TT, Wang YY, Tian YH, Yu JY, Wang D, Xie XLS, Bai F, Wang J. Inferring the evolution and progression of small-cell lung cancer by single-cell sequencing of circulating tumor cells. Clin Cancer Res , 2019, 25(16): 5049-5060. PURPOSE: Genomic analyses of small-cell lung cancer (SCLC) are limited by the availability of tumor specimens. This study aimed to investigate the suitability of single-cell sequencing of circulating tumor cells (CTC) as a method of inferring the evolution and progression of SCLCs. EXPERIMENTAL DESIGN: Between July 1, 2011, and July 28, 2014, 48 consecutively diagnosed patients with SCLC were recruited for this study. CTCs were captured from each patient with CellSearch system. Somatic mutations and copy number alterations (CNA) were monitored by single-cell sequencing of CTCs during chemotherapy. RESULTS: Single-cell sequencing of CTCs can provide a mutational atlas for SCLC. A 10-CNA score based on single CTCs was established as a classifier for outcomes of initial chemotherapy in patients with SCLC. The survival analyses demonstrated that patients with low CNA scores (<0) had significantly prolonged progression-free survival (PFS) and overall survival (OS) after first-line chemotherapy in comparison with those with high scores (>/=0; PFS: 212 days vs. 110.5 days, P = 0.0042; and OS: 223.5 days vs. 424 days, P = 0.0006). The positive predictive value and negative predictive value of the CNA score for clinical subtype (refractory vs. sensitive) were 80.0% and 93.7%, respectively. By tracing allele-specific CNAs in CTCs isolated at different time points during chemotherapy, we showed that CNA heterogeneity might result from allelic losses of initially consistent CNAs. CONCLUSIONS: Single CTC-based sequencing can be utilized to depict the genomic profiles and evolutionary history of SCLC, thus offering the potential for clinical stratification of patients with SCLC.
doi: 10.1158/1078-0432.CCR-18-3571 pmid: 31113842 |
| [45] | Wu BJ, Wang Z, Dong Y, Deng YL, Shi QH. Identification and single-cell sequencing analysis of rare tumor cells in malignant pleural effusion of lung cancer patients. Hereditas(Beijing) , 2019, 41 (2): 175-184. |
| 吴保军, 王卓, 董宇, 邓宇亮, 施奇惠. 肺癌恶性胸腔积液中稀有肿瘤细胞的鉴定与单细胞测序分析. 遗传, 2019, 41 (2): 175-184. | |
| [46] |
Wells D, Escudero T, Levy B, Hirschhorn K, Delhanty JDA, Munné S. First clinical application of comparative genomic hybridization and polar body testing for preimplantation genetic diagnosis of aneuploidy. Fertil Steril , 2002, 78(3): 543-549.
doi: 10.1016/s0015-0282(02)03271-5 pmid: 12215331 |
| [47] | Daina G, Ramos L, Obradors A, Rius M, Martinez- Pasarell O, Polo A, Del Rey J, Obradors J, Benet J, Navarro J. First successful double-factor PGD for lynch syndrome: monogenic analysis and comprehensive aneuploidy screening. Clin Genet , 2013, 84(1): 70-73. |
| [48] |
Tobler KJ, Brezina PR, Benner AT, Du LK, Xu X, Kearns WG. Two different microarray technologies for preimplantation genetic diagnosis and screening, due to reciprocal translocation imbalances, demonstrate equivalent euploidy and clinical pregnancy rates. J Assist Reprod Genet , 2014, 31(7): 843-850.
doi: 10.1007/s10815-014-0230-3 pmid: 24771116 |
| [49] |
Huang J, Yan LY, Fan W, Zhao N, Zhang Y, Tang FC, Xie XS, Qiao J. Validation of multiple annealing and looping- based amplification cycle sequencing for 24-chromosome aneuploidy screening of cleavage-stage embryos. Fertil Steril , 2014, 102(6): 1685-1691. Objective: To validate multiple annealing and looping-based amplification cycle (MALBAC) sequencing for 24-chromosome aneuploidy screening of cleavage embryos and to explore the chromosomal characteristics of embryos at the cleavage stage.Design: The 24-chromosome aneuploidy analyses of the blastomeres included comparative genomic hybridization (CGH), single nucleotide polymorphism (SNP), and MALBAC sequencing.Setting: University-affiliated IVF center.Patient(s): Three couples who delivered babies from the same IVF cycle, which included 23 donated, frozen cleavage embryos.Intervention(s): None.Main Outcome Measure(s): Three blastomeres were selected from each single embryo and subject to CGH, SNP, and MALBAC sequencing for 24-chromosome aneuploidy, respectively. The results of MALBAC sequencing were compared with the results of CGH and SNP. The chromosomal status and occurrence of the abnormal chromosomes were investigated. The relationship between the embryos' morphology and the euploid state was analyzed.Result(s): Among the 23 donated embryos, the MALBAC sequencing results of 18 (78.26%) embryos were identical to those of CGH or SNP, including 8 embryos that had identical results by all three techniques. In terms of euploidy, only 6 of these 23 embryos (26.09%) were diploid. Blastomere abnormality was observed in all autosomes and sex chromosomes. In addition, the frequency of abnormality was different for certain chromosomes.Conclusion(s): With sequencing at 0.04 x genome depth, MALBAC sequencing has been validated as a satisfactory method for 24-chromosome aneuploidy screening. The proportion of abnormal chromosomes was high in cleavage-stage embryos, and any chromosome could be abnormal. (C) 2014 by American Society for Reproductive Medicine.
doi: 10.1016/j.fertnstert.2014.08.015 |
| [50] |
Shang W, Zhang YS, Shu MM, Wang WZ, Ren LK, Chen F, Shao L, Lu SJ, Bo SP, Ma SJ, Gao YM. Comprehensive chromosomal and mitochondrial copy number profiling in human IVF embryos. Reprod Biomed Online , 2018, 36(1): 67-74.
doi: 10.1016/j.rbmo.2017.10.110 pmid: 29203383 |
| [51] |
Macaulay IC, Haerty W, Kumar P, Li YI, Hu TXM, Teng MJ, Goolam M, Saurat N, Coupland P, Shirley LM, Smith M, Van der Aa N, Banerjee R, Ellis PD, Quail MA, Swerdlow HP, Zernicka-Goetz M, Livesey FJ, Ponting CP, Voet T. G&T-seq: parallel sequencing of single-cell genomes and transcriptomes. Nat Methods , 2015, 12(6): 519-522.
doi: 10.1038/nmeth.3370 pmid: 25915121 |
| [52] |
Tang J, Fewings E, Chang D, Zeng HL, Liu SS, Jorapur A, Belote RL, McNeal AS, Tan TM, Yeh I, Arron ST, Judson-Torres RL, Bastian BC, Shain AH. The genomic landscapes of individual melanocytes from human skin. Nature , 2020, 586(7830): 600-605.
doi: 10.1038/s41586-020-2785-8 pmid: 33029006 |
| [53] |
Zhou Y, Bian SH, Zhou X, Cui YL, Wang WD, Wen L, Guo LM, Fu W, Tang FC. Single-cell multiomics sequencing reveals prevalent genomic alterations in tumor stromal cells of human colorectal cancer. Cancer Cell , 2020, 38(6): 818-828.e5.
pmid: 33096021 |
| [54] |
Hou Y, Guo HH, Cao C, Li XL, Hu BQ, Zhu P, Wu XL, Wen L, Tang FC, Huang YY, Peng JR. Single-cell triple omics sequencing reveals genetic, epigenetic, and transcriptomic heterogeneity in hepatocellular carcinomas. Cell Res , 2016, 26(3): 304-319.
doi: 10.1038/cr.2016.23 pmid: 26902283 |
| [55] |
Zachariadis V, Cheng HT, Andrews N, Enge M. A highly scalable method for joint whole-genome sequencing and gene-expression profiling of single cells. Mol Cell , 2020, 80(3): 541-553.e5.
doi: 10.1016/j.molcel.2020.09.025 pmid: 33068522 |
| [1] | Chunhui Ma, Haixu Hu, Lijuan Zhang, Yi Liu, Tianyi Liu. Establishment and verification of a digital PCR assay for the detection of CK19 expression in quantitative analysis of circulating tumor cell [J]. Hereditas(Beijing), 2023, 45(3): 250-260. |
| [2] | Xi Han, Fucheng Luo. Application of single-cell RNA sequencing in probing oligodendroglia heterogeneity and neurological disorders [J]. Hereditas(Beijing), 2023, 45(3): 198-211. |
| [3] | Beiping Zeng, Hongen Xu, Lu Mao, Wenxue Tang. Molecular diagnosis of hereditary deafness and application of stepwise testing strategy [J]. Hereditas(Beijing), 2023, 45(1): 29-41. |
| [4] | Jun Zhou, Chengcheng Zhao, Xiao Wu, Junsong Shi, Rong Zhou, Zhenfang Wu, Zicong Li. Transcriptome heterogeneity of porcine ear fibroblast and its potential influence on embryo development in nuclear transplantation [J]. Hereditas(Beijing), 2020, 42(9): 898-915. |
| [5] | Liang Qu, Su Li, Huaji Qiu. Applications of single-cell RNA sequencing in virology [J]. Hereditas(Beijing), 2020, 42(3): 269-277. |
| [6] | Yaxin Yao,Yongfu La,Ran Di,Qiuyue Liu,Wenping Hu,Xiangyu Wang,Mingxing Chu. Comparison of different single cell whole genome amplification methods and MALBAC applications in assisted reproduction [J]. Hereditas(Beijing), 2018, 40(8): 620-631. |
| [7] | Youwang Lu,Kunhua Wang. Research progress on genetic heterogeneity between primary and paired metastatic colorectal cancer [J]. Hereditas(Beijing), 2017, 39(6): 482-490. |
| [8] | Wenjun Wu, Zhihua Wang, Zhuo Wang, Yuliang Deng, Qihui Shi. Fast isolation and ex vivo culture of circulating tumor cells from the peripheral blood of lung cancer patients [J]. Hereditas(Beijing), 2017, 39(1): 66-74. |
| [9] | Zhen Liu, Jianhong Xu. The application of the high throughput sequencing technology in the transposable elements [J]. HEREDITAS(Beijing), 2015, 37(9): 885-898. |
| [10] | Linqi Wang. Adaptation strategies: how environmental fungi become fatal? [J]. HEREDITAS(Beijing), 2015, 37(5): 436-441. |
| [11] | Shuai Sun, Yuliang Deng. Single-cell detection of EGFR gene mutation in circulating tumor cells in lung cancer [J]. HEREDITAS(Beijing), 2015, 37(12): 1251-1257. |
| [12] | Lu Wen, Fuchou Tang. Recent progress in single-cell RNA-Seq analysis [J]. HEREDITAS(Beijing), 2014, 36(11): 1069-1076. |
| [13] | YANG Yi-Ling CHU Jia-You WANG Ming-Rong. Tumor genetic heterogeneity [J]. HEREDITAS, 2013, 35(1): 1-9. |
| [14] | LI Li, LI Xia~, CHEN Yi-Han, GUO Zheng , JIANG Wei, ZHAGN Rui-Jie, RAO Shao-Qi. Analysis of Diffuse Large B-cell Lymphoma Heterogeneity Based on Coupled Two-way Clustering [J]. HEREDITAS, 2006, 28(9): 1129-1134. |
| [15] | REN Xin-Luan, LI Lian-Xiang, HAN Shu-Ping, CAI Jie, ZHANG Hong-Yi, MA Chang-Yi. Analysis of the Mode of Inheritance in Familial Bronchial Asthma [J]. HEREDITAS, 2006, 28(9): 1067-1070. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||