遗传 ›› 2026, Vol. 48 ›› Issue (2): 201-212.doi: 10.16288/j.yczz.25-234
收稿日期:2025-08-28
修回日期:2025-11-06
出版日期:2025-12-01
发布日期:2025-12-01
通讯作者:
李飞飞,博士,副教授,研究方向:发育与疾病的表观遗传调控。E-mail: liff@scut.edu.cn;作者简介:李雨洁,本科生,专业方向:生物学。E-mail: liyujie_scut@163.com
基金资助:
Yujie Li( ), Feifei Li(
), Feifei Li( ), Wenqing Zhang(
), Wenqing Zhang( ), Qi Chen(
), Qi Chen( )
)
Received:2025-08-28
Revised:2025-11-06
Published:2025-12-01
Online:2025-12-01
Supported by:摘要:
在中枢神经系统稳态维持过程中,小胶质细胞的吞噬功能至关重要。该功能依赖于正常的溶酶体酸化,并由液泡型ATP酶(vacuolar-type ATPase,V-ATPase)精密调控。V-ATPase a3亚基(斑马鱼中由tcirg1b基因编码)的突变是导致人类恶性骨硬化症的主要原因,但其在中枢神经系统中的功能却知之甚少。为了探究V-ATPase a3亚基对斑马鱼中枢神经系统的影响及其作用机制,本研究通过构建tcirg1b基因敲除斑马鱼模型,发现该突变体在胚胎早期未出现神经元发育障碍,但其成鱼脑组织呈现显著病理改变且行为异常。前期数据显示,tcirg1b突变导致小胶质细胞形态胀大,提示其功能可能改变。本研究利用斑马鱼巨噬细胞转录组测序分析发现,吞噬体形成和细胞内pH调节通路基因显著下调。进一步功能验证结果显示,V-ATPase a3亚基缺陷可导致小胶质细胞溶酶体酸化障碍和消化功能受损,继而引发凋亡细胞碎片和TMR-葡聚糖的积累。值得注意的是,当小胶质细胞特异性回补tcirg1b表达时,可成功挽救突变体的行为学表型,提示V-ATPase a3亚基对斑马鱼中枢神经系统的稳态调控可能以小胶质细胞为主体。综上所述,本研究首次在体内证明tcirg1b缺失通过破坏小胶质细胞功能间接导致中枢神经系统稳态失衡,揭示了V-ATPase a3亚基在神经稳态调控中的关键作用,并为相关神经系统疾病的机制研究提供了新的理论依据。
李雨洁, 李飞飞, 张文清, 陈琪. tcirg1b基因缺失导致斑马鱼中枢神经稳态失衡的机制[J]. 遗传, 2026, 48(2): 201-212.
Yujie Li, Feifei Li, Wenqing Zhang, Qi Chen. Mechanism of tcirg1b deficiency in disrupting central nervous system homeostasis of zebrafish[J]. Hereditas(Beijing), 2026, 48(2): 201-212.
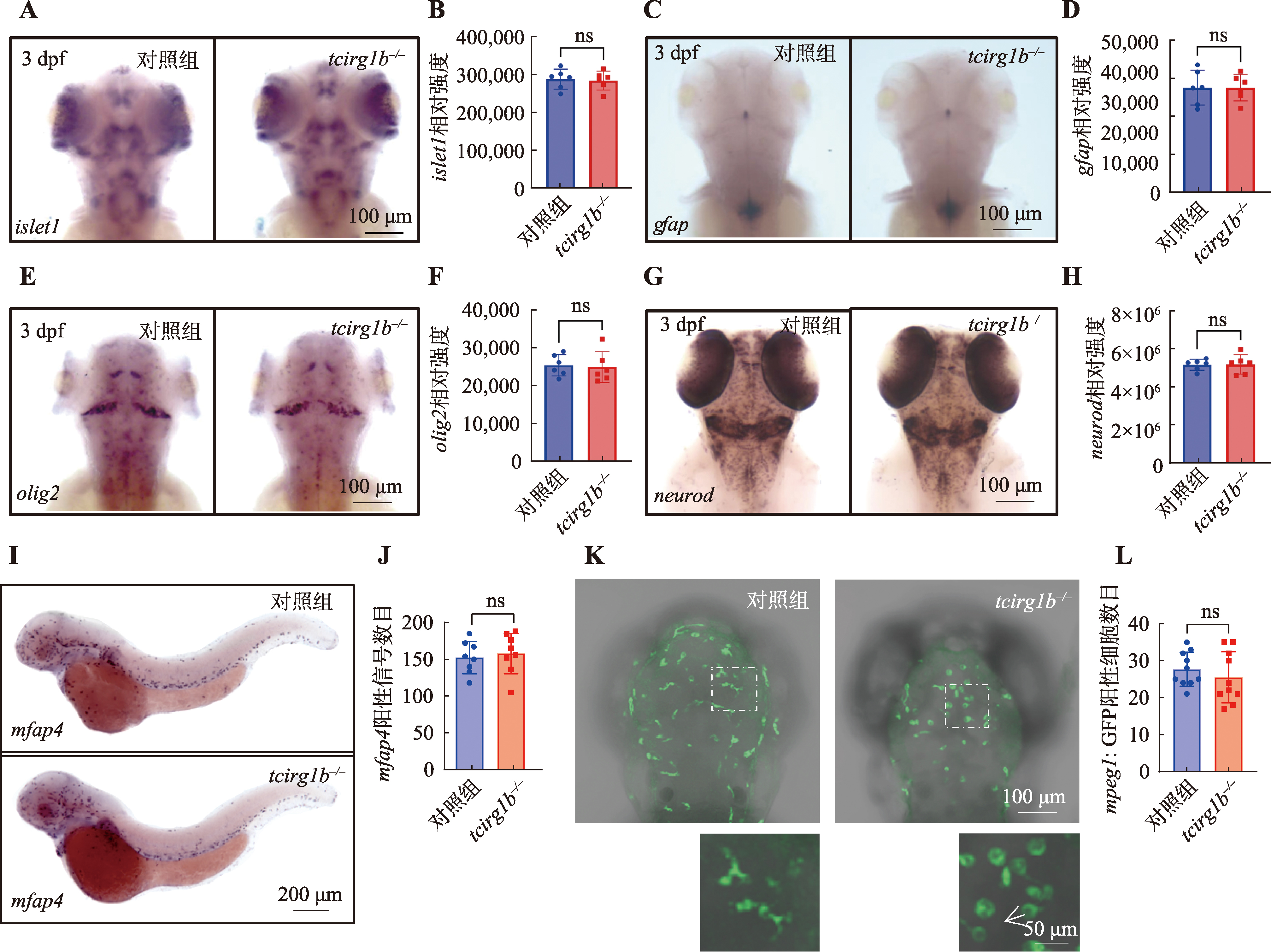
图3
tcirg1b缺陷对斑马鱼早期神经元发育没有明显影响 A和B:3 dpf 对照组和tcirg1b-/-斑马鱼islet1探针原位杂交及其信号的统计分析(每组斑马鱼n=6条);C和D:3 dpf对照组和tcirg1b-/-斑马鱼gfap探针原位杂交及其信号的统计分析(每组斑马鱼n=6条);E和F:3 dpf 对照组和tcirg1b-/-斑马鱼olig2探针原位杂交及其信号的统计分析(每组斑马鱼n =6条);G和H:3 dpf 对照组和tcirg1b-/-斑马鱼neurod探针原位杂交及其信号的统计分析(每组斑马鱼n =6条);I和J:2 dpf对照组和tcirg1b-/-斑马鱼mfap4探针原位杂交及其信号的统计分析(每组斑马鱼n=8条);K和L:Tg(mpeg1:GFP)转基因斑马鱼对照组和tcirg1b-/-脑部的小胶质细胞数目及统计图(白色箭头指示tcirg1b-/-小胶质细胞形态异常;每组斑马鱼n=10条)。ns代表差异不显著(P>0.05),非配对 t 检验,误差线代表Mean±SD。"
| [1] |
Kuan CY, Roth KA, Flavell RA, Rakic P. Mechanisms of programmed cell death in the developing brain. Trends Neurosci, 2000, 23(7): 291-297.
pmid: 10856938 |
| [2] |
Ginhoux F, Lim S, Hoeffel G, Low D, Huber T. Origin and differentiation of microglia. Front Cell Neurosci, 2013, 7: 45.
pmid: 23616747 |
| [3] |
Dos Santos SE, Medeiros M, Porfirio J, Tavares W, Pessôa L, Grinberg L, Leite REP, Ferretti-Rebustini REL, Suemoto CK, Filho WJ, Noctor SC, Sherwood CC, Kaas JH, Manger PR, Herculano-Houzel S. Similar microglial cell densities across brain structures and mammalian species: implications for brain tissue function. J Neurosci, 2020, 40(24): 4622-4643.
pmid: 32253358 |
| [4] |
Daneman R. The blood-brain barrier in health and disease. Ann Neurol, 2012, 72(5): 648-672.
pmid: 23280789 |
| [5] |
Dalmau I, Vela JM, González B, Finsen B, Castellano B. Dynamics of microglia in the developing rat brain. J Comp Neurol, 2003, 458(2): 144-157.
pmid: 12596255 |
| [6] |
Sierra A, Abiega O, Shahraz A, Neumann H. Janus-faced microglia: beneficial and detrimental consequences of microglial phagocytosis. Front Cell Neurosci, 2013, 7: 6.
pmid: 23386811 |
| [7] |
Herzog C, Pons Garcia L, Keatinge M, Greenald D, Moritz C, Peri F, Herrgen L. Rapid clearance of cellular debris by microglia limits secondary neuronal cell death after brain injury in vivo. Development, 2019, 146(9): dev174698.
pmid: 31076485 |
| [8] |
Quick JD, Silva C, Wong JH, Lim KL, Reynolds R, Barron AM, Zeng JL, Lo CH. Lysosomal acidification dysfunction in microglia: an emerging pathogenic mechanism of neuroinflammation and neurodegeneration. J Neuroinflammation, 2023, 20(1): 185.
pmid: 37543564 |
| [9] |
Muench SP, Trinick J, Harrison MA. Structural divergence of the rotary ATPases. Q Rev Biophys, 2011, 44(3): 311-356.
pmid: 21426606 |
| [10] |
Bowman EJ, O'Neill FJ, Bowman BJ. Mutations of pma-1, the gene encoding the plasma membrane H+-ATPase of Neurospora crassa, suppress inhibition of growth by concanamycin A, a specific inhibitor of vacuolar ATPases. J Biol Chem, 1997, 272(23): 14776-14786.
pmid: 9169444 |
| [11] |
Chen FQ, Kang R, Liu J, Tang DL. The V-ATPases in cancer and cell death. Cancer Gene Ther, 2022, 29(11): 1529-1541.
pmid: 35504950 |
| [12] |
Kawasaki-Nishi S, Bowers K, Nishi T, Forgac M, Stevens TH. The amino-terminal domain of the vacuolar proton- translocating ATPase a subunit controls targeting and in vivo dissociation, and the carboxyl-terminal domain affects coupling of proton transport and ATP hydrolysis. J Biol Chem, 2001, 276(50): 47411-47420.
pmid: 11592965 |
| [13] |
Lupanga U, Röhrich R, Askani J, Hilmer S, Kiefer C, Krebs M, Kanazawa T, Ueda T, Schumacher K. The Arabidopsis V-ATPase is localized to the TGN/EE via a seed plant-specific motif. eLife, 2020, 9: e60568.
pmid: 33236982 |
| [14] |
Oka T, Toyomura T, Honjo K, Wada Y, Futai M. Four subunit a isoforms of Caenorhabditis elegans vacuolar H+-ATPase. Cell-specific expression during development. J Biol Chem, 2001, 276(35): 33079-33085.
pmid: 11441002 |
| [15] |
Li YP, Chen W, Liang Y, Li E, Stashenko P. Atp6i- deficient mice exhibit severe osteopetrosis due to loss of osteoclast-mediated extracellular acidification. Nat Genet, 1999, 23(4): 447-451.
pmid: 10581033 |
| [16] | Chu A, Zirngibl RA, Manolson MF. The V-ATPase a3 subunit: structure, function and therapeutic potential of an essential biomolecule in osteoclastic bone resorption. Int J Mol Sci, 2021, 22(13): 6934. |
| [17] |
Sobacchi C, Schulz A, Coxon FP, Villa A, Helfrich MH. Osteopetrosis: genetics, treatment and new insights into osteoclast function. Nat Rev Endocrinol, 2013, 9(9): 522-536.
pmid: 23877423 |
| [18] |
Zhang XY, He JW, Fu WZ, Wang C, Zhang ZL. Novel mutations of TCIRG1 cause a malignant and mild phenotype of autosomal recessive osteopetrosis (ARO) in four Chinese families. Acta Pharmacol Sin, 2017, 38(11): 1456-1465.
pmid: 28816234 |
| [19] | Lin JH, Yang TS, Zhang WQ, Liu W. Role of different Lyl1 transcripts in zebrafish primitive hematopoiesis. Hereditas(Beijing), 2025, 47(5): 573-588. |
| 林杰豪, 杨童舒, 张文清, 刘伟. Lyl1不同转录本在斑马鱼原始造血中的作用. 遗传, 2025, 47(5): 573-588. | |
| [20] |
Thisse C, Thisse B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat Protoc, 2008, 3(1): 59-69.
pmid: 18193022 |
| [21] |
Wu ST, Xue RT, Hassan S, Nguyen TML, Wang TN, Pan HR, Xu J, Liu QF, Zhang WQ, Wen ZL. Il34-Csf1r pathway regulates the migration and colonization of microglial precursors. Dev Cell, 2018, 46(5): 552-563.e4.
pmid: 30205037 |
| [22] |
Chen Q, Kou HJ, Demy DL, Liu W, Li JC, Wen ZL, Herbomel P, Huang ZB, Zhang WQ, Xu J. The different roles of V-ATPase a subunits in phagocytosis/endocytosis and autophagy. Autophagy, 2024, 20(10): 2297-2313.
pmid: 38873931 |
| [23] |
Zhang ML, Li XP, Gao LF, Liu J, Bi ZJ, Miao YH, Shan Y, Yu HL. Nobiletin, an activator of the pyruvate kinase isozyme M1/M2 protein, upregulated the glycolytic signalling pathway and alleviated depressive-like behaviour caused by artificial light exposure at night in zebrafish. Food Chem, 2025, 463(Pt 2): 141328.
pmid: 39305673 |
| [24] |
Raj B, Wagner DE, McKenna A, Pandey S, Klein AM, Shendure J, Gagnon JA, Schier AF. Simultaneous single-cell profiling of lineages and cell types in the vertebrate brain. Nat Biotechnol, 2018, 36(5): 442-450.
pmid: 29608178 |
| [25] |
Vidal-Itriago A, Radford RAW, Aramideh JA, Maurel C, Scherer NM, Don EK, Lee A, Chung RS, Graeber MB, Morsch M. Microglia morphophysiological diversity and its implications for the CNS. Front Immunol, 2022, 13: 997786.
pmid: 36341385 |
| [26] |
Westman J, Walpole GFW, Kasper L, Xue BY, Elshafee O, Hube B, Grinstein S. Lysosome fusion maintains phagosome integrity during fungal infection. Cell Host Microbe, 2020, 28(6): 798-812.e6.
pmid: 33022213 |
| [27] |
Chen YP, Kou YH, Ni Y, Yang HT, Xu CL, Fan HG, Liu HQ. Microglia efferocytosis: an emerging mechanism for the resolution of neuroinflammation in Alzheimer's disease. J Neuroinflammation, 2025, 22(1): 96.
pmid: 40159486 |
| [28] |
Borst K, Dumas AA, Prinz M. Microglia: immune and non-immune functions. Immunity, 2021, 54(10): 2194-2208.
pmid: 34644556 |
| [29] |
Guzmán-Ruíz MA, Guerrero Vargas NN, Ramírez-Carreto RJ, González-Orozco JC, Torres-Hernández BA, Valle- Rodríguez M, Guevara-Guzmán R, Chavarría A. Microglia in physiological conditions and the importance of understanding their homeostatic functions in the arcuate nucleus. Front Immunol, 2024, 15: 1392077.
pmid: 39295865 |
| [30] | Yin J, Valin KL, Dixon ML, Leavenworth JW. The role of microglia and macrophages in CNS homeostasis, autoimmunity, and cancer. J Immunol Res, 2017, 5150678. |
| [31] |
Serra EL, Medalha CC, Mattioli R. Natural preference of zebrafish (Danio rerio) for a dark environment. Braz J Med Biol Res, 1999, 32(12): 1551-1553.
pmid: 10585639 |
| [32] |
Scimeca JC, Franchi A, Trojani C, Parrinello H, Grosgeorge J, Robert C, Jaillon O, Poirier C, Gaudray P, Carle GF. The gene encoding the mouse homologue of the human osteoclast-specific 116-kDa V-ATPase subunit bears a deletion in osteosclerotic (oc/oc) mutants. Bone, 2000, 26(3): 207-213.
pmid: 10709991 |
| [33] |
Falace A, Volpedo G, Scala M, Zara F, Striano P, Fassio A. V-ATPase dysfunction in the brain: genetic insights and therapeutic opportunities. Cells, 2024, 13(17): 1441.
pmid: 39273013 |
| [34] |
Aoto K, Kato M, Akita T, Nakashima M, Mutoh H, Akasaka N, Tohyama J, Nomura Y, Hoshino K, Ago Y, Tanaka R, Epstein O, Ben-Haim R, Heyman E, Miyazaki T, Belal H, Takabayashi S, Ohba C, Takata A, Mizuguchi T, Miyatake S, Miyake N, Fukuda A, Matsumoto N, Saitsu H. ATP6V0A1 encoding the a1-subunit of the V0 domain of vacuolar H+-ATPases is essential for brain development in humans and mice. Nat Commun, 2021, 12(1): 2107.
pmid: 33833240 |
| [35] |
Bagh MB, Peng SY, Chandra G, Zhang ZJ, Singh SP, Pattabiraman N, Liu AY, Mukherjee AB. Misrouting of v-ATPase subunit V0a1 dysregulates lysosomal acidification in a neurodegenerative lysosomal storage disease model. Nat Commun, 2017, 8(1): 14612.
pmid: 28266544 |
| [36] |
Koçak G, Güzel BN, Mıhçı E, Küpesiz OA, Yalçın K, Manguoğlu AE. TCIRG1 and SNX10 gene mutations in the patients with autosomal recessive osteopetrosis. Gene, 2019, 702: 83-88.
pmid: 30898715 |
| [37] |
Goldmann T, Wieghofer P, Jordão MJC, Prutek F, Hagemeyer N, Frenzel K, Amann L, Staszewski O, Kierdorf K, Krueger M, Locatelli G, Hochgerner H, Zeiser R, Epelman S, Geissmann F, Priller J, Rossi FMV, Bechmann I, Kerschensteiner M, Linnarsson S, Jung S, Prinz M. Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat Immunol, 2016, 17(7): 797-805.
pmid: 27135602 |
| [38] |
Gao C, Jiang JW, Tan YY, Chen SD. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduct Target Ther, 2023, 8(1): 359.
pmid: 37735487 |
| [39] |
Brockerhoff SE, Dowling JE, Hurley JB. Zebrafish retinal mutants. Vision Res, 1998, 38(10): 1335-1339.
pmid: 9667001 |
| [40] |
Gestri G, Link BA, Neuhauss SCF. The visual system of zebrafish and its use to model human ocular diseases. Dev Neurobiol, 2012, 72(3): 302-327.
pmid: 21595048 |
| [41] |
Martin A, Babbitt A, Pickens AG, Pickett BE, Hill JT, Suli A. Single-cell RNA sequencing characterizes the molecular heterogeneity of the larval zebrafish optic tectum. Front Mol Neurosci, 2022, 15: 818007.
pmid: 35221915 |
| [42] |
Tesmer AL, Fields NP, Robles E. Input from torus longitudinalis drives binocularity and spatial summation in zebrafish optic tectum. BMC Biol, 2022, 20(1): 24.
pmid: 35073895 |
| [43] |
Walton ZE, Brooks RC, Dang CV. mTOR senses intracellular pH through lysosome dispersion from RHEB. Bioessays, 2019, 41(7): e1800265.
pmid: 31157925 |
| [44] |
Yambire KF, Rostosky C, Watanabe T, Pacheu-Grau D, Torres-Odio S, Sanchez-Guerrero A, Senderovich O, Meyron-Holtz EG, Milosevic I, Frahm J, West AP, Raimundo N. Impaired lysosomal acidification triggers iron deficiency and inflammation in vivo. eLife, 2019, 8: e51031.
pmid: 31793879 |
| [45] |
Zhang WJ, Bai JW, Hang K, Xu JX, Zhou CW, Li LJ, Wang ZX, Wang YB, Wang KB, Xue DT. Role of lysosomal acidification dysfunction in mesenchymal stem cell senescence. Front Cell Dev Biol, 2022, 10: 817877.
pmid: 35198560 |
| [1] | 郑少辉, 刘洋, 夏新欣, 刘彦梅. 碱基编辑技术及其在斑马鱼中的开发应用[J]. 遗传, 2026, 48(1): 46-60. |
| [2] | 高云海, 邓家杰, 肖霄, 潘鲁湲, 何牡丹, 张蕴斌. 斑马鱼ppp6r3调控性腺分化和配子发生的作用研究[J]. 遗传, 2025, 47(9): 1023-1031. |
| [3] | 林杰豪, 杨童舒, 张文清, 刘伟. Lyl1不同转录本在斑马鱼原始造血中的作用[J]. 遗传, 2025, 47(5): 573-588. |
| [4] | 寇涵婧, 黄志斌, 张文清, 陈琪. V-ATPase a3亚基调控小胶质细胞吞噬体成熟的机制[J]. 遗传, 2025, 47(11): 1256-1268. |
| [5] | 刘吉祥, 赖思婷, 白晶, 徐进. Il34拯救甲硝唑导致的斑马鱼中枢神经系统轴突再生障碍[J]. 遗传, 2024, 46(6): 478-489. |
| [6] | 洪佳馨, 徐颂恩, 张文清, 刘伟. Pu.1和cMyb在斑马鱼中性粒细胞发育中的相互作用[J]. 遗传, 2024, 46(4): 319-332. |
| [7] | 孙飘, 李颖, 刘帆, 王璐. TPI缺乏症斑马鱼模型的构建及分析[J]. 遗传, 2024, 46(3): 232-241. |
| [8] | 杨晓君, 黄振瀚, 刘伟, 张文清, 黄志斌. CD209同源基因在斑马鱼中的鉴定及功能表征[J]. 遗传, 2024, 46(11): 947-957. |
| [9] | 李凯伦, 卢荆奥, 陈小辉, 张文清, 刘伟. 尿囊素促进破骨细胞缺陷斑马鱼骨折修复[J]. 遗传, 2023, 45(4): 341-353. |
| [10] | 商晓康, 张思萌, 倪军军. 组织蛋白酶B参与脑衰老及阿尔兹海默症发生发展研究进展[J]. 遗传, 2023, 45(3): 212-220. |
| [11] | 卢荆澳, 黄春燕, 林芷茵, 唐政, 马宁, 黄志斌. cd99l2基因调控斑马鱼白细胞组织间的迁移机制[J]. 遗传, 2022, 44(9): 798-809. |
| [12] | 郑鹏飞, 谢海波, 朱盼盼, 赵呈天. 斑马鱼神经底板处神经元的分布及特征[J]. 遗传, 2022, 44(6): 510-520. |
| [13] | 张婷婷, 刘峰. 斑马鱼蛋白酪氨酸硫酸化修饰的检测方法研究[J]. 遗传, 2022, 44(2): 178-186. |
| [14] | 贾婷婷, 雷蕾, 吴歆媛, 蔡顺有, 陈艺璇, 薛钰. 二甲双胍对斑马鱼骨骼发育及损伤修复的机制研究[J]. 遗传, 2022, 44(1): 68-79. |
| [15] | 郭佳妮, 刘帆, 王璐. 斑马鱼血液疾病模型及应用[J]. 遗传, 2020, 42(8): 725-738. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||

www.chinagene.cn
备案号:京ICP备09063187号-4
总访问:,今日访问:,当前在线:
