遗传 ›› 2026, Vol. 48 ›› Issue (2): 177-200.doi: 10.16288/j.yczz.25-205
江山1,2,3( ), 雷宇航1,2,3(
), 雷宇航1,2,3( ), 廖天赐1,2,3, 钟易1,2,3, 甘麦邻1,2,3, 朱砺1,2,3, 沈林園1,2,3(
), 廖天赐1,2,3, 钟易1,2,3, 甘麦邻1,2,3, 朱砺1,2,3, 沈林園1,2,3( )
)
收稿日期:2025-08-04
修回日期:2025-09-09
出版日期:2026-02-20
发布日期:2025-11-04
通讯作者:
沈林園,博士,副教授,研究方向:猪的分子遗传与育种、猪生物育种技术研发与应用。E-mail: shenlinyuan@sicau.edu.cn作者简介:江山,本科生,专业方向:动物遗传育种与繁殖。E-mail: 202200429@stu.sicau.edu.cn基金资助:
Shan Jiang1,2,3( ), Yuhang Lei1,2,3(
), Yuhang Lei1,2,3( ), Tianci Liao1,2,3, Yi Zhong1,2,3, Mailin Gan1,2,3, Li Zhu1,2,3, Linyuan Shen1,2,3(
), Tianci Liao1,2,3, Yi Zhong1,2,3, Mailin Gan1,2,3, Li Zhu1,2,3, Linyuan Shen1,2,3( )
)
Received:2025-08-04
Revised:2025-09-09
Published:2026-02-20
Online:2025-11-04
Supported by:摘要:
红肉与白肉的健康效应争议长期存在,传统基于动物种属的二元分类体系难以匹配其营养异质性与疾病风险的复杂性。本文通过整合流行病学、分子机制及食品科学多维证据,系统重构了肉类健康评估框架。研究证实,传统红白肉定义因忽视种内差异及跨物种悖论,需转向基于部位、加工方式的精准分类;加工肉作为1 类致癌物与未加工红肉风险存在本质差异,后者适量摄入有助于平衡血红素铁/维生素B12的营养价值与相关健康风险;在致病机制上,红肉中血红素铁、Neu5Gc、TMAO及加工衍生的NOCs、HCAs/PAHs构成多重病理网络;而白肉因缺失核心毒性成分且富含ω-3 PUFA(EPA/DHA抗炎与神经保护),多数研究提示可能会降低心血管代谢风险,但高温加工可削弱其优势。基于此,国际指南形成“严控加工肉、限量未加工红肉、优选白肉”共识,并发展基因筛查、益生菌调控菌群-TMAO轴及低温烹饪等精准策略。未来需突破单细胞多组学解析器官特异性损伤、建立基因-菌群-营养素三元交互的个体化风险评估模型,并通过CRISPR育种靶向降低红肉毒性及AI优化膳食推荐系统,推动肉类消费向营养-健康-环境可持续性协同进化。
江山, 雷宇航, 廖天赐, 钟易, 甘麦邻, 朱砺, 沈林園. 红肉与白肉健康效应的多学科证据评估与机制整合[J]. 遗传, 2026, 48(2): 177-200.
Shan Jiang, Yuhang Lei, Tianci Liao, Yi Zhong, Mailin Gan, Li Zhu, Linyuan Shen. Multidimensional evidence assessment and mechanistic integration of the health effects of red and white meat[J]. Hereditas(Beijing), 2026, 48(2): 177-200.
表1
各类红白肉7种营养元素含量表"
| 类别 | 肉类种类 | 营养元素 | ||||||
|---|---|---|---|---|---|---|---|---|
| 铁 (mg) | 总脂肪 (g) | 饱和脂肪 (g) | 胆固醇(mg) | PUFAs (g) | EPA (g) | DHA (g) | ||
| 红肉 | 碎牛肉 | 2.88 | 4.46 | 2.210 | 88 | 0.281 | 0 | 0 |
| 侧腹牛排 | 1.80 | 9.31 | 3.840 | 81 | 0.390 | 0.003 | 0.001 | |
| 碎猪肉 | 1.05 | 7.15 | 1.950 | 78 | 0.776 | 0.003 | 0.004 | |
| 猪里脊肉 | 0.96 | 17.60 | 6.220 | 84 | 0.700 | 0 | 0 | |
| 白肉 | 火鸡腿肉 | 2.30 | 9.82 | 3.060 | 85 | 2.720 | 0 | 0.050 |
| 鸡腿肉 | 1.40 | 8.06 | 2.200 | 89 | 1.880 | 0.010 | 0.050 | |
| 蓝鳍金枪鱼 | 1.31 | 6.28 | 1.610 | 49 | 1.840 | 0.363 | 1.140 | |
| 鳕鱼 | 0.59 | 1.26 | 0.170 | 91 | 0.108 | 0.021 | 0.059 | |
| 虾 | 0.51 | 0.28 | 0.056 | 189 | 0.079 | 0.015 | 0.015 | |
| 鸡胸肉 | 0.43 | 3.61 | 1.190 | 99 | 0.660 | 0.010 | 0.020 | |
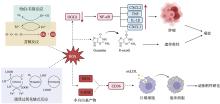
图1
血红素铁致病机制示意图 血红素铁通过芬顿反应和哈伯-韦斯反应催化活性氧(ROS)的生成。由此产生的ROS,特别是高活性的·OH,可通过两条主要途径诱导细胞损伤。首先,·OH可直接攻击DNA,导致8-oxoG的形成及随后的遗传毒性。这种DNA损伤还能通过调节OGG1和激活NF-κB来触发炎症级联反应,上调促炎细胞因子(TNF、IL-1β、CXCL1、CXCL2),从而促进肿瘤发生和癌症发展。其次,ROS可引发脂质过氧化,这是一个产生MDA和4-HNE等活性醛类的链式反应。这些副产物通过上调清道夫受体CD36,促进巨噬细胞对oxLDL的摄取,导致泡沫细胞的形成,最终促进动脉粥样硬化的病理进程。"
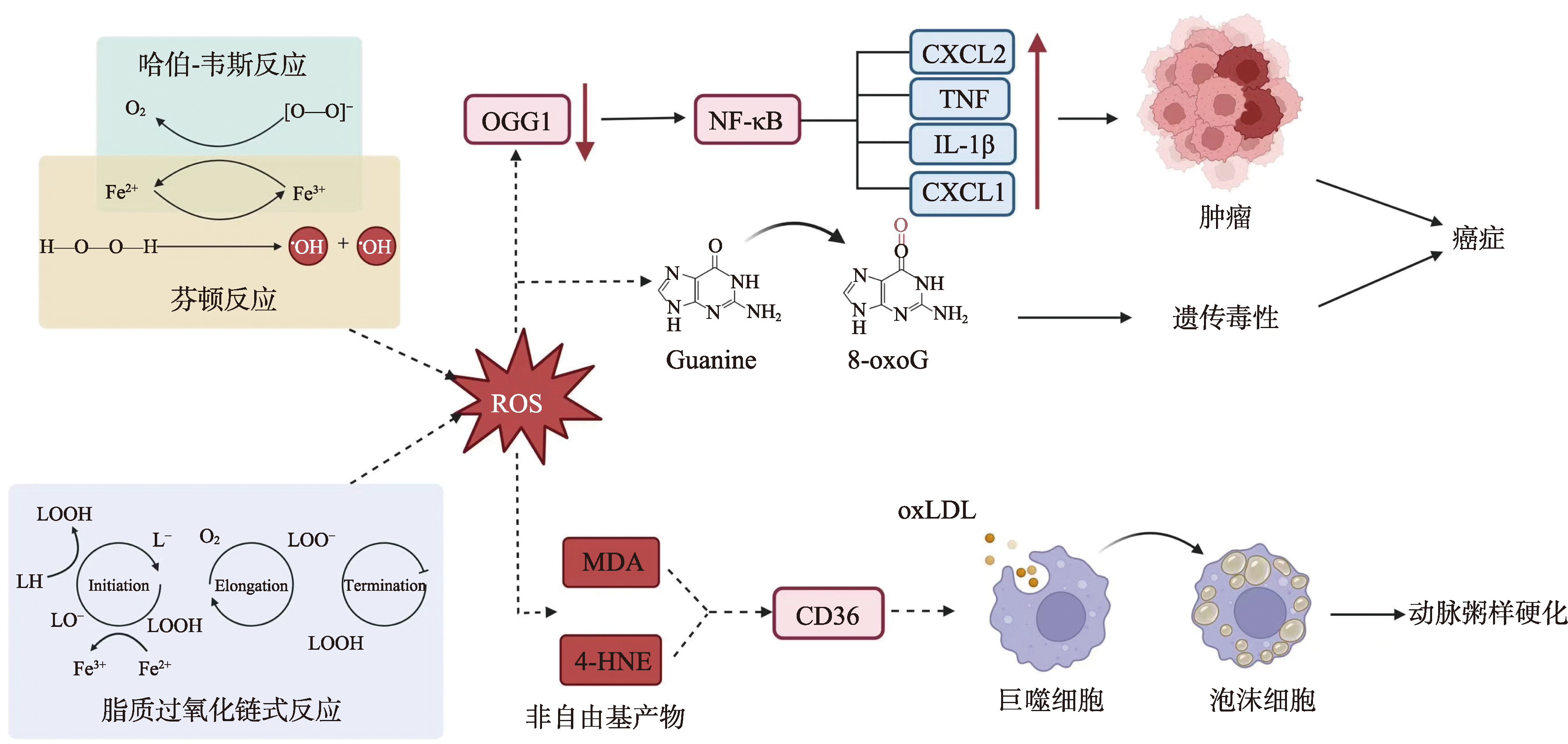
图2
晚期糖基化终产物(AGEs)致病机制 在高温烹饪红肉过程中产生的AGEs,在与其受体结合后,会启动多种致病信号级联反应。AGE-RAGE的相互作用通过激活NADPH氧化酶以及一个涉及RAC1和c-Src的DIAPH1依赖性途径,触发ROS的产生。这种氧化应激促进了动脉粥样硬化的发展。同时,AGE-RAGE轴激活多个下游通路,包括Ras-ERK1/2和PKC/MAPKs,这些通路共同作用于转录因子NF-κB。活化的NF-κB转移至细胞核,并上调多种促炎和粘附分子基因(如MCP-1、TNF-α、IL-6、VCAM-1、ICAM-1)的表达,从而促进炎症、癌症和肾脏疾病。此外,AGE-RAGE的结合可诱导内质网应激,抑制Slc2a4基因转录,导致GLUT4蛋白水平降低。由此引起的葡萄糖摄取障碍导致高血糖,并促进了糖尿病的发病。"
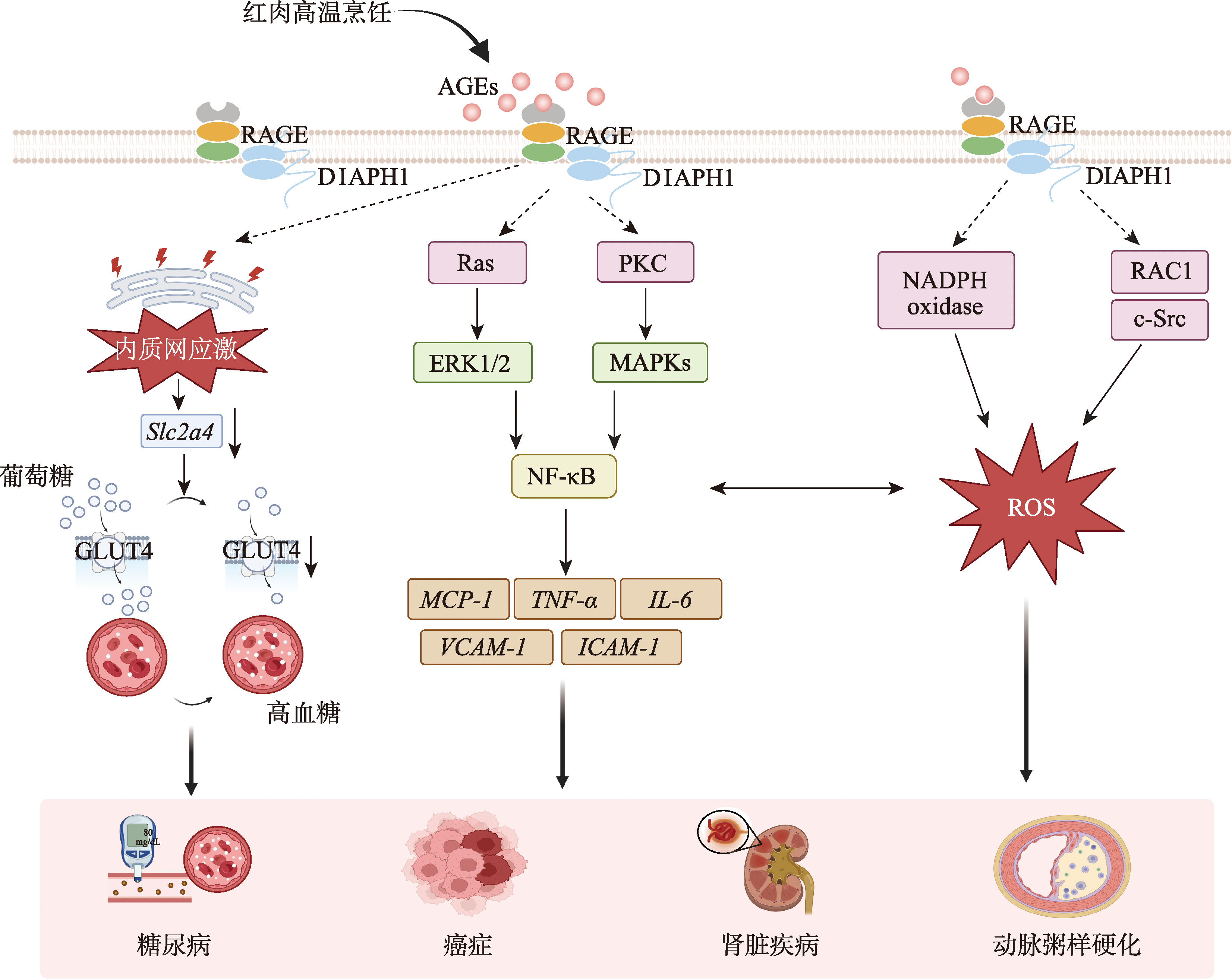
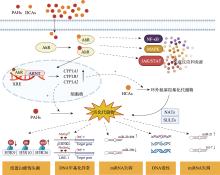
图3
多环芳烃(PAHs)和杂环胺类化合物(HCAs)致病机制 在经过细胞摄取后,PAHs和HCAs均可与AhR结合。活化的AhR-配体复合物转移到细胞核,与ARNT二聚化,并结合到靶基因启动子区域的XRE,显著上调细胞色素P450酶基因(CYP1A1、CYP1B1、CYP1A2)。这些酶分别将PAHs和HCAs代谢为高活性中间体和环外氨基羟基化代谢物。HCAs的代谢物可被NATs或SULTs进一步活化,形成亲电物质,这些物质易与DNA加合,导致DNA毒性和链断裂。除了直接的DNA损伤,这些活性代谢物还会诱导一系列表观遗传改变,包括组蛋白修饰失衡(如H3K9乙酰化、H3S10磷酸化、H3K36三甲基化的改变)、DNA甲基化异常(如p16INK4的高甲基化、LINE-1的低甲基化)和miRNA失调(如miR-22/494和miR-21的上调,miR-29b和let-7家族的下调)。这些表观遗传修饰共同导致基因组不稳定和基因表达异常,从而驱动癌变。此外,PAHs和HCAs通过AhR激活,通过调节NF-κB、MAPK和JAK/STAT等关键信号通路,触发免疫反应和炎症,加剧细胞损伤并促进疾病进展。"
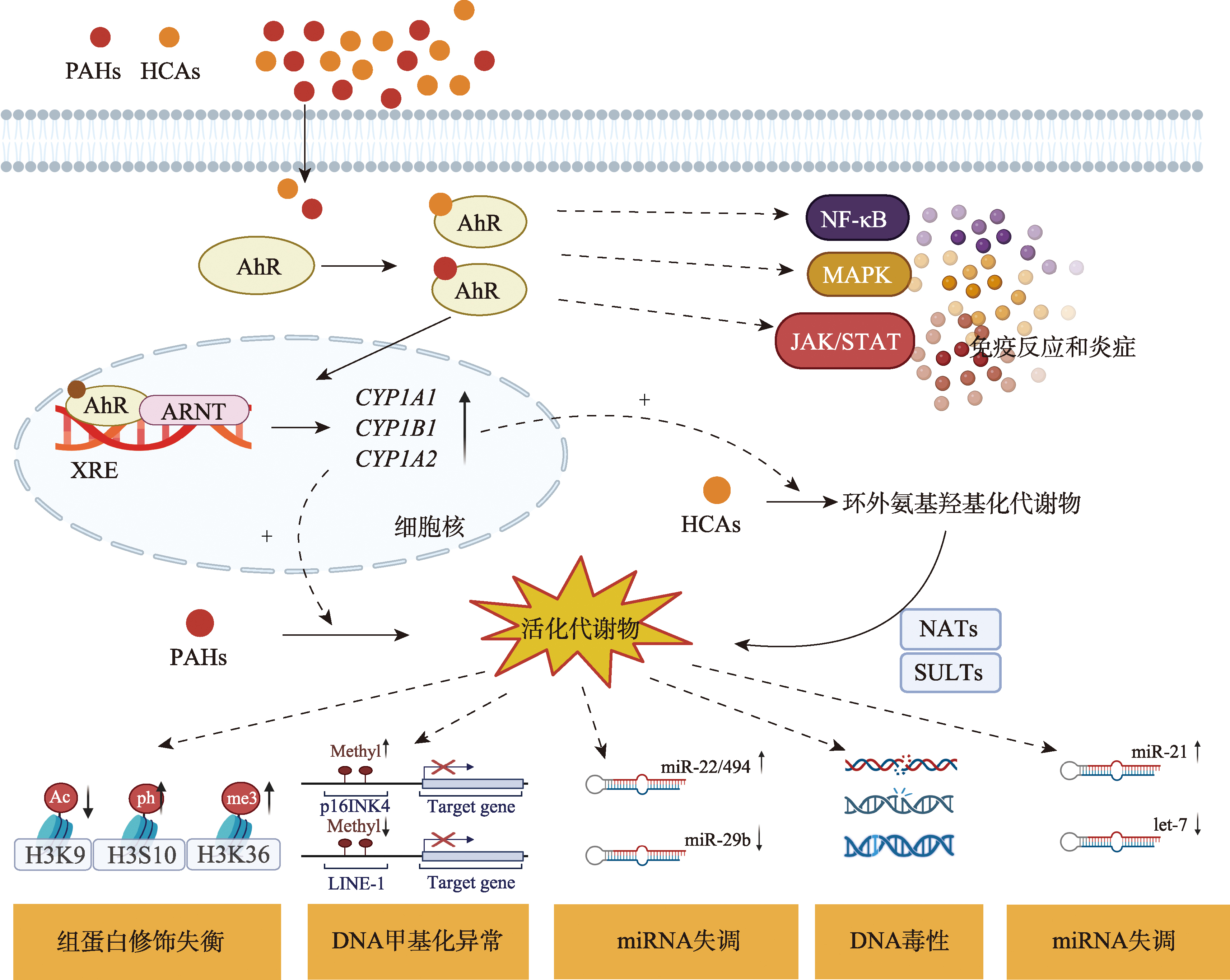
图4
N-羟乙醇酰神经氨酸(Neu5Gc)致病机制示意图 膳食中的Neu5Gc是一种富含于红肉中的外源性唾液酸,通过多种信号通路发挥其促癌作用,尤其是在结直肠癌中。它通过结合HRAS和SRC等靶点直接激活PI3K-Akt通路,导致AKT2磷酸化以及细胞周期调节因子(CDK2、CCNA2)的上调,从而加速细胞周期进程并促进细胞增殖。此外,Neu5Gc增强Wnt3a配体-受体结合,激活Wnt通路,导致β-catenin积累和下游基因(AXIN2、MYC)表达增加,从而促进结直肠癌细胞增殖。除了致癌信号,Neu5Gc还通过IκB-α和P65磷酸化激活NF-κB通路,导致ROS产生增加和炎症,从而促进炎症性肠病。同时,Neu5Gc显著降低紧密连接蛋白(ZO-1、occludin、claudin-1、claudin-4)的表达,损害肠上皮屏障的完整性,促进病原体和细菌的易位。此外,Neu5Gc诱导肠道菌群失调,表现为有益菌(如Muribaculaceae、Lachnospiraceae、Prevotellaceae)减少,促炎菌(如Bacteroidetes、Firmicutes、Prevotellaceae_NK3B31_group、Clostridia UCG-014)增加,进一步激活NF-κB通路并加剧慢性炎症。"
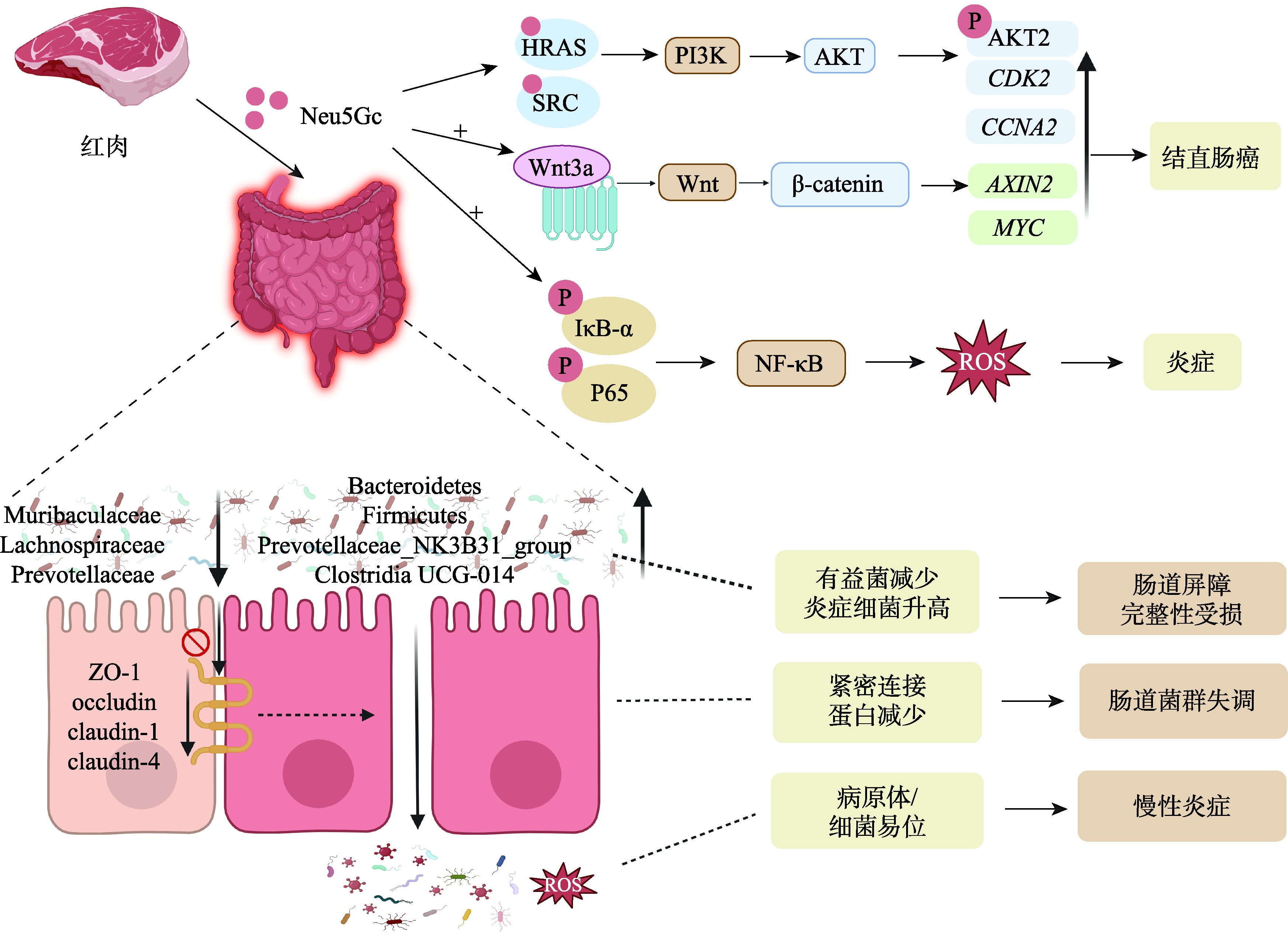
图5
氧化三甲胺(TMAO)致病机制示意图 人体摄入红肉后,胆碱和左旋肉碱被肠道微生物代谢为TMA,TMA随后在肝脏中被FMOs氧化为TMAO。全身TMAO水平升高通过促进SERCA2a与自噬蛋白ATG5的相互作用,导致SERCA2a降解和随后的钙稳态失衡,从而导致心肌肥厚。在动脉粥样硬化中,TMAO通过MAPK/JNK通路,上调清道夫受体(特别是CD36),增强巨噬细胞泡沫细胞的形成,从而增加oxLDL的摄取。此外,TMAO扰乱胆汁酸代谢,改变血清胆汁酸谱(如DCA升高,TMCA降低),从而激活核受体FXR和SHP。这一级联反应抑制Cyp7a1的表达,减少胆汁酸合成并损害RCT,最终加速动脉粥样硬化斑块的进展。在胃肠道中,TMAO通过下调紧密连接蛋白(ZO-1、occludin),激活TLR4/MyD88/NF-κB炎症通路,并抑制Wnt/β-catenin通路,从而损害肠道屏障完整性,导致肠道通透性增加以及细菌/毒素易位。同时,TMAO通过诱导肝窦内皮细胞毛细血管化(CD31、vWF表达上调;eNOS表达下调),促进纤维化基因(α-SMA、TIMP1)表达,并将巨噬细胞极化偏向促炎M1表型(TNF-α、IL-1β、IL-6增加;IL-10减少),从而导致非酒精性脂肪肝(NAFLD),共同加剧肝脏炎症和纤维化。"
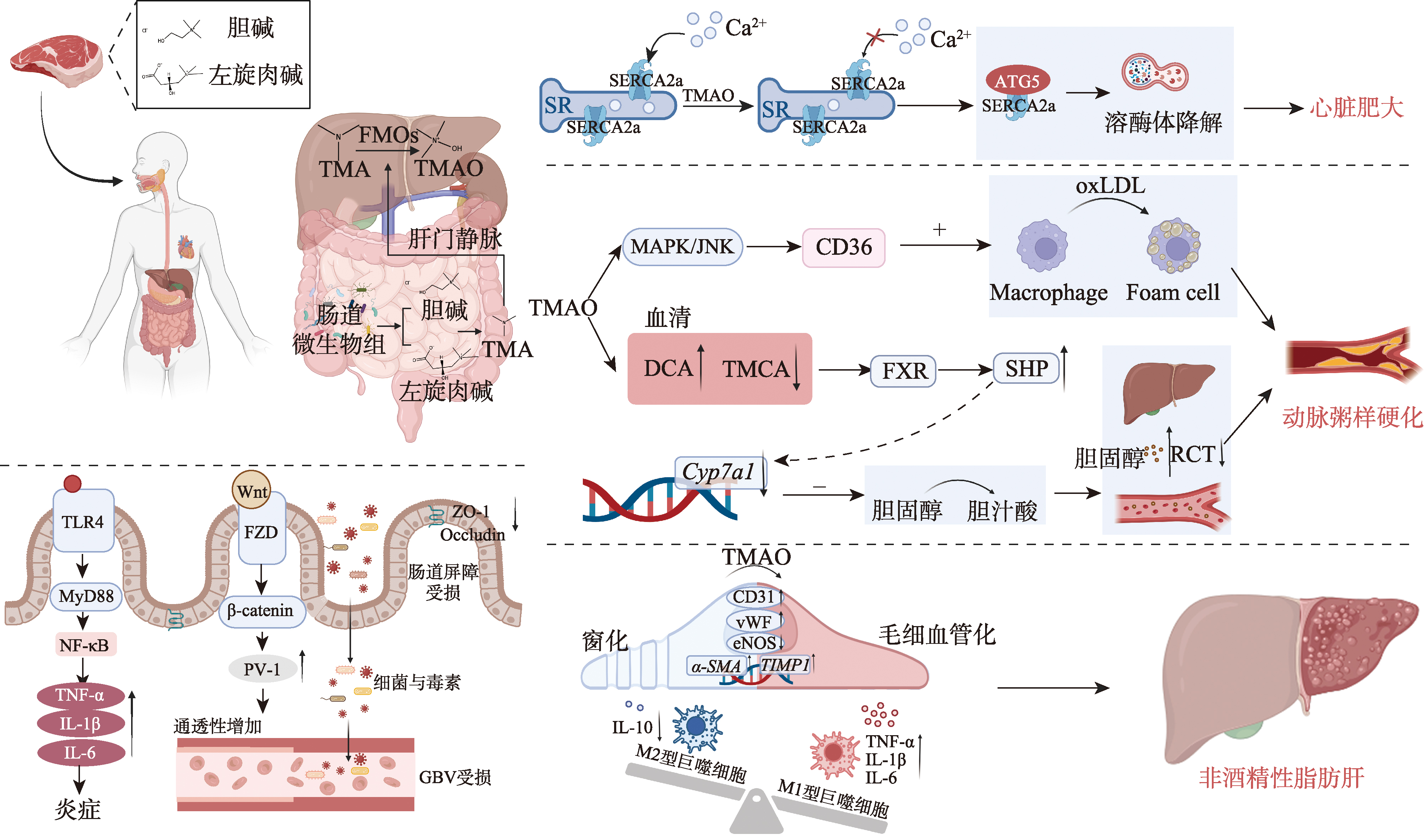
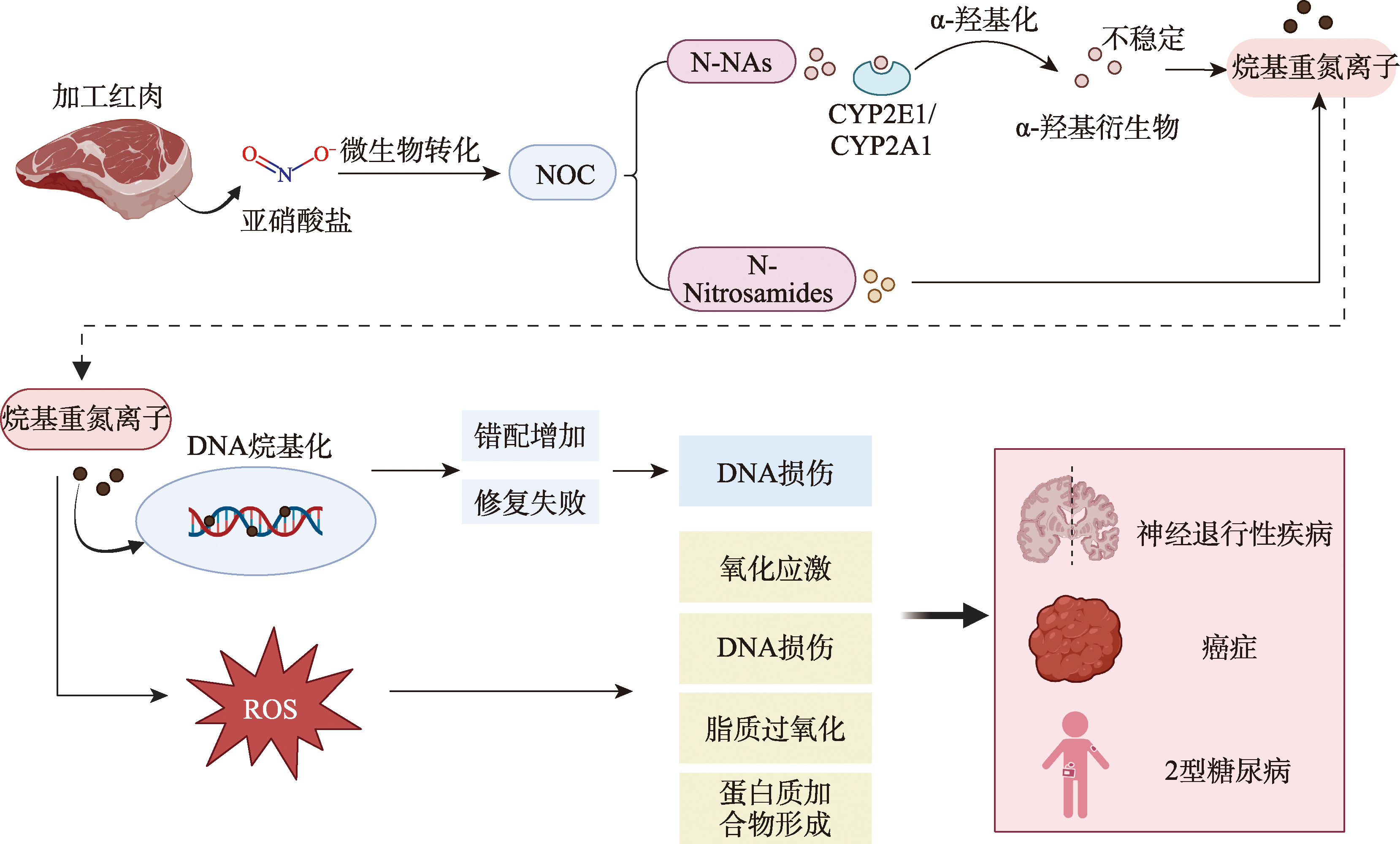
图6
N-亚硝基化合物(NOCs)致病机制示意图 NOCs由加工肉类中的亚硝酸盐经微生物生物转化形成,是一类重要的遗传毒性物质。NOCs主要包括N-NAs和N-亚硝酰胺两大类。N-NAs(如NDMA和NDEA),主要通过细胞色素P450酶(CYP2E1/CYP2A1)进行α-羟基化代谢活化,生成不稳定的α-羟基衍生物,这些衍生物自发分解为高活性的烷基重氮离子。相反,N-亚硝酰胺无需代谢活化即可直接烷基化DNA。这两种途径最终都导致DNA烷基化,形成DNA加合物(如O⁶-烷基鸟嘌呤、N⁷-烷基鸟嘌呤),增加错配,并损害DNA修复机制,最终导致DNA损伤。此外,NOCs的代谢活化会产生ROS,从而诱导氧化应激、进一步的DNA损伤、脂质过氧化和蛋白质加合物形成。这些细胞损伤通过激活促炎细胞因子(IL-1β、IL-6、TNF-α)和促进胰岛素抵抗,共同导致多种慢性疾病的发生,包括神经退行性疾病、癌症和2型糖尿病。"
| [1] | Geiker NRW, Bertram HC, Mejborn H, Dragsted LO, Kristensen L, Carrascal JR, Bügel S, Astrup A. Meat and human health—current knowledge and research gaps. Foods, 2021, 10(7): 1556. |
| [2] | Rohrmann S, Overvad K, Bueno-de-Mesquita HB, Jakobsen MU, Egeberg R, Tjønneland A, Nailler L, Boutron-Ruault MC, Clavel-Chapelon F, Krogh V, Palli D, Panico S, Tumino R, Ricceri F, Bergmann MM, Boeing H, Li KR, Kaaks R, Khaw KT, Wareham NJ, Crowe FL, Key TJ, Naska A, Trichopoulou A, Trichopoulos D, Leenders M, Peeters PHM, Engeset D, Parr CL, Skeie G, Jakszyn P, Sánchez MJ, Huerta JM, Redondo ML, Barricarte A, Amiano P, Drake I, Sonestedt E, Hallmans G, Johansson I, Fedirko V, Romieux I, Ferrari P, Norat T, Vergnaud AC, Riboli E, Linseisen J. Meat consumption and mortality—results from the european prospective investigation into cancer and nutrition. BMC Med, 2013, 11(1): 63. |
| [3] |
Iqbal R, Dehghan M, Mente A, Rangarajan S, Wielgosz A, Avezum A, Seron P, AlHabib KF, Lopez-Jaramillo P, Swaminathan S, Mohammadifard N, Zatońska K, Bo H, Varma RP, Rahman O, Yusufali A, Lu Y, Ismail N, Rosengren A, Imeryuz N, Yeates K, Chifamba J, Dans A, Kumar R, Liu X, Tsolekile L, Khatib R, Diaz R, Teo K, Yusuf S. Associations of unprocessed and processed meat intake with mortality and cardiovascular disease in 21 countries [Prospective Urban Rural Epidemiology (PURE) Study]: a prospective cohort study. Am J Clin Nutr, 2021, 114(3): 1049-1058.
pmid: 33787869 |
| [4] |
Steinberg P. Red meat-derived nitroso compounds, lipid peroxidation products and colorectal cancer. Foods, 2019, 8(7): 252.
pmid: 31336781 |
| [5] | Adeyeye SAO, Sivapriya T, Sankarganesh P. Formation and mitigation of heterocyclic amines (HCAs) and polycyclic aromatic hydrocarbons (PAHs) in high temperature processed meat products: a review. Discover Food, 2025, 5(1): 258. |
| [6] | Rot T, Kovačević D, Habschied K, Mastanjević K. N-nitrosamines in meat products: formation, detection and regulatory challenges. Processes, 2025, 13(5): 1555. |
| [7] |
Wu GY. Important roles of dietary taurine, creatine, carnosine, anserine and 4-hydroxyproline in human nutrition and health. Amino Acids, 2020, 52(3): 329-360.
pmid: 32072297 |
| [8] | Jairath G, Biswas AK, Mal G, Suman SP. Bioactive compounds in meat: their roles in modulating palatability and nutritional value. Meat Muscle Biol, 2024, 8(1): 1-15. |
| [9] |
Goethals S, Vossen E, Michiels J, Vanhaecke L, Van Camp J, Van Hecke T, De Smet S. Impact of red versus white meat consumption in a prudent or western dietary pattern on the oxidative status in a pig model. J Agric Food Chem, 2019, 67(19): 5661-5671.
pmid: 31012581 |
| [10] |
Li J, Li YT, Feng SF, He KY, Guo LLZ, Chen WW, Wang M, Zhong LX, Wu CT, Peng XJ, Tang SH. Differential effects of dietary white meat and red meat on NAFLD progression by modulating gut microbiota and metabolites in rats. Oxid Med Cell Longev, 2022, 2022: 6908934.
pmid: 36035222 |
| [11] | Q&A on the carcinogenicity of the consumption of red meat and processed meat. International Agency for Research on Cancer, 2015. |
| [12] |
Weng KQ, Huo WR, Li Y, Zhang Y, Zhang Y, Chen GH, Xu Q. Fiber characteristics and meat quality of different muscular tissues from slow- and fast-growing broilers. Poult Sci, 2022, 101(1): 101537.
pmid: 34788716 |
| [13] | Keeton JT, Dikeman ME. ‘Red’ and ‘white’ meats— terms that lead to confusion. Anim Front, 2017, 7(4): 29-33. |
| [14] | Fischer J. Veg or non-veg? From bazaars to hypermarkets in India. Int J Asia Pac Stud, 2019, 15(1): 1-32. |
| [15] |
Guasch-Ferré M, Willett WC. The Mediterranean diet and health: a comprehensive overview. J Intern Med, 2021, 290(3): 549-566.
pmid: 34423871 |
| [16] | Seman DL, Boler DD, Carr CC, Dikeman ME, Owens CM, Keeton JT, Pringle TD, Sindelar JJ, Woerner DR, De Mello AS, Powell TH. Meat science lexicon. Meat Muscle Biol, 2018, 2(3): 127-141. |
| [17] |
Pearce N, Blair A, Vineis P, Ahrens W, Andersen A, Anto JM, Armstrong BK, Baccarelli AA, Beland FA, Berrington A, Bertazzi PA, Birnbaum LS, Brownson RC, Bucher JR, Cantor KP, Cardis E, Cherrie JW, Christiani DC, Cocco P, Coggon D, Comba P, Demers PA, Dement JM, Douwes J, Eisen EA, Engel LS, Fenske RA, Fleming LE, Fletcher T, Fontham E, Forastiere F, Frentzel-Beyme R, Fritschi L, Gerin M, Goldberg M, Grandjean P, Grimsrud TK, Gustavsson P, Haines A, Hartge P, Hansen J, Hauptmann M, Heederik D, Hemminki K, Hemon D, Hertz-Picciotto I, Hoppin JA, Huff J, Jarvholm B, Kang D, Karagas MR, Kjaerheim K, Kjuus H, Kogevinas M, Kriebel D, Kristensen P, Kromhout H, Laden F, Lebailly P, LeMasters G, Lubin JH, Lynch CF, Lynge E, Mannetje A, McMichael AJ, McLaughlin JR, Marrett L, Martuzzi M, Merchant JA, Merler E, Merletti F, Miller A, Mirer FE, Monson R, Nordby KC, Olshan AF, Parent ME, Perera FP, Perry MJ, Pesatori AC, Pirastu R, Porta M, Pukkala E, Rice C, Richardson DB, Ritter L, Ritz B, Ronckers CM, Rushton L, Rusiecki JA, Rusyn I, Samet JM, Sandler DP, de Sanjose S, Schernhammer E, Costantini AS, Seixas N, Shy C, Siemiatycki J, Silverman DT, Simonato L, Smith AH, Smith MT, Spinelli JJ, Spitz MR, Stallones L, Stayner LT, Steenland K, Stenzel M, Stewart BW, Stewart PA, Symanski E, Terracini B, Tolbert PE, Vainio H, Vena J, Vermeulen R, Victora CG, Ward EM, Weinberg CR, Weisenburger D, Wesseling C, Weiderpass E, Zahm SH. IARC monographs:40 years of evaluating carcinogenic hazards to humans. Environ Health Perspect, 2015, 123(6): 507-514.
pmid: 25712798 |
| [18] | Frank SM, Taillie LS, Jaacks LM. How Americans eat red and processed meat: an analysis of the contribution of thirteen different food groups. Public Health Nutr, 2022, 25(5): 1406-1415. |
| [19] |
Phillips JA. Dietary guidelines for americans,2020-2025. Workplace Health Saf, 2021, 69(8): 395.
pmid: 34279148 |
| [20] | Chinese Residents' Dietary Guidelines (2022) Released in Beijing. Acta Nutrimenta Sinica, 2022, 44(6): 521-522. |
| 《中国居民膳食指南(2022)》在京发布. 营养学报, 2022, 44(6): 521-522. | |
| [21] | Ling ZY, Liu JN. Research progress on Mediterranean diet and health. Chin J Urban Rural Enterp Health, 2024, 39(2): 18-21. |
| 凌子羽, 刘菊凝. 地中海饮食与健康研究进展. 中国城乡企业卫生, 2024, 39(2): 18-21. | |
| [22] |
Rossi L, Ferrari M, Ghiselli A. The alignment of recommendations of dietary guidelines with sustainability aspects: lessons learned from Italy’s example and proposals for future development. Nutrients, 2023, 15(3): 542.
pmid: 36771249 |
| [23] |
de Carvalho AM, de Castro Selem SS, Miranda AM, Marchioni DM. Excessive red and processed meat intake: relations with health and environment in Brazil. Br J Nutr, 2016, 115(11): 2011-2016.
pmid: 27040312 |
| [24] | Ministry of Health of Brazil. Dietary guidelines for the brazilian population. 2015. |
| [25] |
Du HD, Guo Y, Bennett DA, Bragg F, Bian Z, Chadni M, Yu CQ, Chen YP, Tan YL, Millwood IY, Gan W, Yang L, Yao P, Luo GJ, Li JG, Qin YL, Lv J, Lin X, Key T, Chen JS, Clarke R, Li LM, Chen ZM, China Kadoorie Biobank collaborative group. Red meat, poultry and fish consumption and risk of diabetes: a 9 year prospective cohort study of the China kadoorie biobank. Diabetologia, 2020, 63(4): 767-779.
pmid: 31970429 |
| [26] |
Zheng Y, Li YP, Satija A, Pan A, Sotos-Prieto M, Rimm E, Willett WC, Hu FB. Association of changes in red meat consumption with total and cause specific mortality among US women and men: two prospective cohort studies. BMJ, 2019, 365: l2110.
pmid: 31189526 |
| [27] |
Sinha R, Cross AJ, Graubard BI, Leitzmann MF, Schatzkin A. Meat intake and mortality: a prospective study of over half a million people. Arch Intern Med, 2009, 169(6): 562-571.
pmid: 19307518 |
| [28] |
Etemadi A, Sinha R, Ward MH, Graubard BI, Inoue-Choi M, Dawsey SM, Abnet CC. Mortality from different causes associated with meat, heme iron, nitrates, and nitrites in the NIH-AARP diet and health study: population based cohort study. BMJ, 2017, 357: j1957.
pmid: 28487287 |
| [29] | Nagao M, Iso H, Yamagishi K, Date C, Tamakoshi A. Meat consumption in relation to mortality from cardiovascular disease among japanese men and women. Eur J Clin Nutr, 2012, 66(6): 687-693. |
| [30] | Buerkli S, Pei SN, Hsiao SC, Lee CT, Zeder C, Zimmermann MB, Moretti D. The TMPRSS6 variant (SNP rs855791) affects iron metabolism and oral iron absorption-a stable iron isotope study in Taiwanese women. Haematologica, 2021, 106(11): 2897-2905. |
| [31] |
Tang N, Chen LQ, Zhuang H. Effects of heme iron enriched peptide on iron deficiency anemia in rats. Food Funct, 2014, 5(2): 390-399.
pmid: 24326613 |
| [32] |
Hunt JR. Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. Am J Clin Nutr, 2003, 78(3 Suppl): 633S-639S.
pmid: 12936958 |
| [33] |
Ekmekcioglu C, Wallner P, Kundi M, Weisz U, Haas W, Hutter HP. Red meat, diseases, and healthy alternatives: a critical review. Crit Rev Food Sci Nutr, 2018, 58(2): 247-261.
pmid: 27128451 |
| [34] |
Davis H, Magistrali A, Butler G, Stergiadis S. Nutritional benefits from fatty acids in organic and grass-fed beef. Foods, 2022, 11(5): 646.
pmid: 35267281 |
| [35] |
Constante M, Fragoso G, Calvé A, Samba-Mondonga M, Santos MM. Dietary heme induces gut dysbiosis, aggravates colitis, and potentiates the development of adenomas in mice. Front Microbiol, 2017, 8: 1809.
pmid: 28983289 |
| [36] |
Sajadimajd S, Khazaei M. Oxidative stress and cancer: the role of Nrf2. Curr Cancer Drug Targets, 2018, 18(6): 538-557.
pmid: 28969555 |
| [37] |
Demarquoy J. Revisiting the role of carnitine in heart disease through the lens of the gut microbiota. Nutrients, 2024, 16(23): 4244.
pmid: 39683637 |
| [38] |
Jing LL, Zhang HH, Xiang QN, Shen L, Guo XX, Zhai CL, Hu HL. Targeting trimethylamine N-oxide: a new therapeutic strategy for alleviating atherosclerosis. Front Cardiovasc Med, 2022, 9: 864600.
pmid: 35770223 |
| [39] |
Thøgersen R, Gray N, Kuhnle G, Van Hecke T, De Smet S, Young JF, Sundekilde UK, Hansen AK, Bertram HC. Inulin-fortification of a processed meat product attenuates formation of nitroso compounds in the gut of healthy rats. Food Chem, 2020, 302: 125339.
pmid: 31419771 |
| [40] |
Yavari M, Kalupahana NS, Harris BN, Ramalingam L, Zu YJ, Kahathuduwa CN, Moustaid-Moussa N. Mechanisms linking obesity, insulin resistance, and alzheimer’s disease: effects of polyphenols and omega-3 polyunsaturated fatty acids. Nutrients, 2025, 17(7): 1203.
pmid: 40218960 |
| [41] | Wagan G, Bakhsh L, Mehmood A, Bano S, Memon S, Samo RP. Effects of red meat consumption on histology of coronary arteries in adult albino mice. Ann Punjab Med Coll, 2019, 13(4), 287-291. |
| [42] | Yang WH, Liu C, Han NX, Zhang P. Research progress on the safety of meat curing process. Meat Ind, 2022, (11): 39-45. |
| 杨文鹤, 刘策, 韩乃瑄, 张沛. 肉类腌制工艺安全性的研究进展. 肉类工业, 2022, (11): 39-45. | |
| [43] | Xie YF, Geng YQ, Yao JB, Ji JF, Chen F, Xiao JB, Hu XS, Ma LJ. N-nitrosamines in processed meats: Exposure, formation and mitigation strategies. J Agric Food Res, 2023, 13: 100645. |
| [44] |
Racovita RC, Secuianu C, Ciuca MD, Israel-Roming F. Effects of smoking temperature, smoking time, and type of wood sawdust on polycyclic aromatic hydrocarbon accumulation levels in directly smoked pork sausages. J Agric Food Chem, 2020, 68(35): 9530-9536.
pmid: 32786847 |
| [45] |
Chiang VSC, Quek SY. The relationship of red meat with cancer: Effects of thermal processing and related physiological mechanisms. Crit Rev Food Sci Nutr, 2017, 57(6): 1153-1173.
pmid: 26075652 |
| [46] | Qi DS, Zhang Q. Progress in research on formation mechanism and control methods for acrylamide and heterocyclic amines during food frying. Food Sci, 2021, 42(21): 338-346. |
| 戚岱莎, 张清. 食物油炸过程中丙烯酰胺和杂环胺的形成及控制方法研究进展. 食品科学, 2021, 42(21): 338-346. | |
| [47] | Xu WG, Sun XM, Liu W. The influence of modern cooking techniques on the nutritional value of traditional food. China Food Saf Mag, 2024, (33): 98-100. |
| 许文广, 孙小明, 刘薇. 现代烹饪技术对传统食品营养价值的影响. 食品安全导刊, 2024, (33): 98-100. | |
| [48] |
Gibis M. Heterocyclic aromatic amines in cooked meat products: causes, formation, occurrence, and risk assessment. Compr Rev Food Sci Food Saf, 2016, 15(2): 269-302.
pmid: 33371602 |
| [49] |
Yao GM, Zhou YJ, Li ZP, Ma QS. Heterocyclic aromatic amines in roasted chicken: formation and prediction based on heating temperature and time. Food Chem, 2023, 405(Pt A): 134822.
pmid: 36371838 |
| [50] |
Tan K, Lim L, Peng Y, Cheong KL. Effects of food processing on the lipid nutritional quality of commercially important fish and shellfish. Food Chem X, 2023, 20: 101034.
pmid: 38144794 |
| [51] | Malesza IJ, Bartkowiak-Wieczorek J, Winkler-Galicki J, Nowicka A, Dzięciołowska D, Błaszczyk M, Gajniak P, Słowińska K, Niepolski L, Walkowiak J, Mądry E. The dark side of iron: the relationship between iron, inflammation and gut microbiota in selected diseases associated with iron deficiency anaemia—a narrative review. Nutrients, 2022, 14(17): 3478. |
| [52] |
Lu YF, Li T, Shu Y, Lu CY, Luo ZQ, Wang JR, Xiong H, Li WY. Lipid peroxidation and sarcopenia: molecular mechanisms and potential therapeutic approaches. Front Med (Lausanne), 2025, 12: 1525205.
pmid: 39963429 |
| [53] |
Huang YX, Sarkhel S, Roy A, Mohan A. Interrelationship of lipid aldehydes (MDA, 4-HNE, and 4-ONE) mediated protein oxidation in muscle foods. Crit Rev Food Sci Nutr, 2024, 64(32): 11809-11825.
pmid: 37589270 |
| [54] |
Yun MR, Im DS, Lee SJ, Park HM, Bae SS, Lee WS, Kim CD. 4-hydroxynonenal enhances CD36 expression on murine macrophages via p38 MAPK-mediated activation of 5-lipoxygenase. Free Radic Biol Med, 2009, 46(5): 692-698.
pmid: 19135147 |
| [55] |
Mushenkova NV, Bezsonov EE, Orekhova VA, Popkova TV, Starodubova AV, Orekhov AN. Recognition of oxidized lipids by macrophages and its role in atherosclerosis development. Biomedicines, 2021, 9(8): 915.
pmid: 34440119 |
| [56] | Halliwell B, Adhikary A, Dingfelder M, Dizdaroglu M. Hydroxyl radical is a significant player in oxidative DNA damage in vivo. Chem Soc Rev, 2021, 50(15): 8355-8360. |
| [57] | Zhao Y, Wang CX, Yang TM, Li CS, Zhang LH, Du DN, Wang RX, Wang J, Wei M, Ba XQ. Linking oxidative DNA lesion 8-OxoG to tumor development and progression. Hereditas(Beijing), 2022, 44(6): 466-477. |
| 赵岩, 王晨鑫, 杨天明, 李春爽, 张丽宏, 杜冬妮, 王若曦, 王静, 魏民, 巴雪青. DNA氧化损伤8-羟鸟嘌呤与肿瘤的发生发展. 遗传, 2022, 44(6): 466-477. | |
| [58] |
Han X, Shi ZH, Wu Z, Zeng XQ, Sun YY, Yao KY, Shen QW, Fan XK, Luo J, Pan DD. AGEs in cooked meat: production, detection, and mechanisms of its inhibition by plant extracts. Food Res Int, 2025, 207: 116067.
pmid: 40086958 |
| [59] |
Yue Q, Song Y, Liu Z, Zhang L, Yang L, Li JL. Receptor for advanced glycation end products (RAGE): a pivotal hub in immune diseases. Molecules, 2022, 27(15): 4922.
pmid: 35956875 |
| [60] |
Senatus LM, Schmidt AM. The AGE-RAGE axis: implications for age-associated arterial diseases. Front Genet, 2017, 8: 187.
pmid: 29259621 |
| [61] |
Suzuki S, Hayashi T, Egawa T. Advanced glycation end products promote ROS production via PKC/p47 phox axis in skeletal muscle cells. J Physiol Sci, 2024, 74(1): 51.
pmid: 39369187 |
| [62] |
Fukami K, Yamagishi SI, Okuda S. Role of AGEs-RAGE system in cardiovascular disease. Curr Pharm Des, 2014, 20(14): 2395-2402.
pmid: 23844818 |
| [63] | Wu X, Li N, Xu HD, Li M. Research progress in the formation and inhibition mechanism of advanced glycation end products. Food Sci, 2023, 44(17): 204-214. |
| 吴旋, 李娜, 徐怀德, 李梅. 晚期糖基化终末产物形成及抑制机理的研究进展. 食品科学, 2023, 44(17): 204-214. | |
| [64] |
Passarelli M, Machado UFF. AGEs-induced and endoplasmic reticulum stress/inflammation-mediated regulation of GLUT4 expression and atherogenesis in diabetes mellitus. Cells, 2021, 11(1): 104.
pmid: 35011666 |
| [65] |
Xu XX, Liu XF, Zhang JX, Liang L, Wen CT, Li YD, Shen MY, Wu YY, He XD, Liu GY, Xu X. Formation, migration, derivation, and generation mechanism of polycyclic aromatic hydrocarbons during frying. Food Chem, 2023, 425: 136485.
pmid: 37276667 |
| [66] |
Du W, Jiang S, Lei YL, Wang JZ, Cui ZP, Xiang P, Chang ZF, Duan WY, Shen GF, Qin YM, Pan B, Yu YJ. Occurrence, formation mechanism, and health risk of polycyclic aromatic hydrocarbons in barbecued food. Ecotoxicol Environ Saf, 2025, 293: 118046.
pmid: 40086033 |
| [67] |
Stading R, Gastelum G, Chu C, Jiang WW, Moorthy B. Molecular mechanisms of pulmonary carcinogenesis by polycyclic aromatic hydrocarbons (PAHs): implications for human lung cancer. Semin Cancer Biol, 2021, 76: 3-16.
pmid: 34242741 |
| [68] |
Das DN, Ravi N. Influences of polycyclic aromatic hydrocarbon on the epigenome toxicity and its applicability in human health risk assessment. Environ Res, 2022, 213: 113677.
pmid: 35714684 |
| [69] | Lin ZY, Pang TY, Wu JW, Jin H. Role of polycyclic aromatic hydrocarbons in the occurrence and development of inflammatory skin diseases. Chin J Dermatol, 2024, 57(8): 765-769. |
| 林子沅, 庞天怡, 武静文, 靳慧. 多环芳烃在炎症性皮肤病发生发展中的作用研究进展. 中华皮肤科杂志, 2024, 57(8): 765-769. | |
| [70] |
Bahman F, Choudhry K, Al-Rashed F, Al-Mulla F, Sindhu S, Ahmad R. Aryl hydrocarbon receptor: current perspectives on key signaling partners and immunoregulatory role in inflammatory diseases. Front Immunol, 2024, 15: 1421346.
pmid: 39211042 |
| [71] |
Knize MG, Felton JS. Formation and human risk of carcinogenic heterocyclic amines formed from natural precursors in meat. Nutr Rev, 2005, 63(5): 158-165.
pmid: 15971410 |
| [72] | Liu GY, Wu YY, Xu XW, Xu XX, Liang L, Zhang JX, Wen CT, Li YD, He XD, Xu X, Liu XF. The relationship between the deterioration of frying oil and the generation of hazards during frying. Food Addit Contam: Part A, 2024, 41(12): 1554-1569. |
| [73] | Xue GZ, Huang XQ, Song LJ, Qiao MW, Zhao JS, Ma XJ, Xu JT. Progress in the prevention of the formation of heterocyclic amines in high-temperature meat products and the regulation of their metabolism in the human body. Food Sci, 2022, 43(13): 256-266. |
| 薛桂中, 黄现青, 宋莲军, 乔明武, 赵建生, 马相杰, 徐俊涛. 高温肉制品杂环胺防控及体内代谢调控研究进展. 食品科学, 2022, 43(13): 256-266. | |
| [74] | Wang HT, Sun XY, Wang D, Guo J, Jin Y, Zhao LH. Research progress of formation and control measures of heterocyclic aromatic amines in meat products. Food Res Dev, 2022, 43(5): 195-203. |
| 王惠汀, 孙学颖, 王丹, 郭进, 靳烨, 赵丽华. 肉制品中杂环胺类化合物形成及控制措施的研究进展. 食品研究与开发, 2022, 43(5): 195-203. | |
| [75] |
Chen YS, Wang R, Dashwood WM, Löhr CV, Williams DE, Ho E, Mertens-Talcott S, Dashwood RH. A miRNA signature for an environmental heterocyclic amine defined by a multi-organ carcinogenicity bioassay in the rat. Arch Toxicol, 2017, 91(10): 3415-3425.
pmid: 28289824 |
| [76] |
Sekimoto M, Sumi H, Hosaka T, Umemura T, Nishikawa A, Degawa M. Aryl hydrocarbon receptor activation and CYP1A induction by cooked food-derived carcinogenic heterocyclic amines in human HepG2 cell lines. Food Chem Toxicol, 2016, 97: 256-264.
pmid: 27644595 |
| [77] | Altman MO, Gagneux P. Absence of Neu5Gc and presence of anti-Neu5Gc antibodies in humans—an evolutionary perspective. Front Immunol, 2019, 10: 789. |
| [78] |
Guo YX, Ren HL, Wang H, Xiao YR, Wang C, Liu MD, Duan FC, Li HS, Hu P, Li YS, Liu ZS, Lu SY. Characterization of an aptamer targeting Neu5Gc, as an endogenous pathogenic factor derived from red meat. Molecules, 2024, 29(6): 1273.
pmid: 38542909 |
| [79] | Zhang LC, Liu YN, La XQ, Yan SN, Chen Y, Liang JY, Li ZY. The potential mechanism of Neu5Gc inducing colorectal cancer based on network pharmacology and experimental validation. Naunyn-Schmiedeberg's Arch Pharmacol, 2023, 396(4): 705-718. |
| [80] |
Lopes AL, Paulino AC, Thaumaturgo MAS, Araújo WM, Caloba P, Kawanishi K, Willert K, De Oliveira RP, Machado JC, Lemos F, Varki N, Morgado-Diaz JA, Läubli H, Todeschini AR, Varki A, Alisson-Silva F. Dietary intake of the red meat-derived glycan Neu5Gc fuels colorectal cancer through up-regulation of Wnt signaling pathway. Cancer Lett, 2025, 616: 217598.
pmid: 40023392 |
| [81] |
He EQ, Quan W, Luo J, Liu CX, Zheng WT, Shen QW. Absorption and transport mechanism of red meat-derived N-glycolylneuraminic acid and its damage to intestinal barrier function through the NF-κB signaling pathway. Toxins (Basel), 2023, 15(2): 132.
pmid: 36828446 |
| [82] | He LL, Liu CX, Xiao Y, Shen QW, Wu YY. Proinflammatory mechanism of N-glycolylneuraminic acid in mice through regulating the gut microbiota. Food Sci, 2024, 45(14): 103-113. |
| 贺乐丽, 刘楚鑫, 肖毅, 沈清武, 吴艳阳. N-羟乙酰神经氨酸通过调节肠道菌群致小鼠的炎症机制. 食品科学, 2024, 45(14): 103-113. | |
| [83] | Krüger R, Merz B, Rist MJ, Ferrario PG, Bub A, Kulling SE, Watzl B. Associations of current diet with plasma and urine TMAO in the KarMeN study: direct and indirect contributions. Mol Nutr Food Res, 2017, 61(11): 1700363. |
| [84] |
Gatarek P, Kaluzna-Czaplinska J. Trimethylamine N-oxide (TMAO) in human health. EXCLI J, 2021, 20: 301-319.
pmid: 33746664 |
| [85] |
Wang ZN, Bergeron N, Levison BS, Li XS, Chiu S, Jia X, Koeth RA, Li L, Wu YP, Tang WHW, Krauss RM, Hazen SL. Impact of chronic dietary red meat, white meat, or non-meat protein on trimethylamine N-oxide metabolism and renal excretion in healthy men and women. Eur Heart J, 2019, 40(7): 583-594.
pmid: 30535398 |
| [86] |
Thomas MS, Fernandez ML. Trimethylamine N-oxide (TMAO), diet and cardiovascular disease. Curr Atheroscler Rep, 2021, 23(4): 12.
pmid: 33594574 |
| [87] |
de Quadros Iorra F, Rodrigues PG, Bock PM, Guahnon MP, Eller S, de Oliveira TF, Birk L, de Souza Schwarz P, Drehmer M, Bloch KV, Cureau FV, Schaan BD. Gut microbiota metabolite TMAO and adolescent cardiometabolic health: a cross-sectional analysis. J Endocr Soc, 2025, 9(5): bvaf055.
pmid: 40242209 |
| [88] |
Chistiakov DA, Melnichenko AA, Myasoedova VA, Grechko AV, Orekhov AN. Mechanisms of foam cell formation in atherosclerosis. J Mol Med (Berl), 2017, 95(11): 1153-1165.
pmid: 28785870 |
| [89] |
Wang ZN, Klipfell E, Bennett BJ, Koeth R, Levison BS, DuGar B, Feldstein AE, Britt EB, Fu XM, Chung YM, Wu YP, Schauer P, Smith JD, Allayee H, Tang WHW, DiDonato JA, Lusis AJ, Hazen SL. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature, 2011, 472(7341): 57-63.
pmid: 21475195 |
| [90] |
Geng J, Yang CC, Wang BJ, Zhang XW, Hu TT, Gu Y, Li J. Trimethylamine N-oxide promotes atherosclerosis via CD36-dependent MAPK/JNK pathway. Biomed Pharmacother, 2018, 97: 941-947.
pmid: 29136772 |
| [91] | Ouimet M, Barrett TJ, Fisher EA. HDL and reverse cholesterol transport: basic mechanisms and their roles in vascular health and disease. Circ Res, 2019, 124(10): 1505-1518. |
| [92] |
Ding L, Chang MR, Guo Y, Zhang LY, Xue CH, Yanagita T, Zhang TT, Wang YM. Trimethylamine-N-oxide (TMAO)-induced atherosclerosis is associated with bile acid metabolism. Lipids Health Dis, 2018, 17(1): 286.
pmid: 30567573 |
| [93] |
Lei DY, Liu Y, Liu Y, Jiang YJ, Lei YY, Zhao FL, Li WQ, Ouyang ZH, Chen LL, Tang SY, Ouyang DS, Li XH, Li Y. The gut microbiota metabolite trimethylamine N-oxide promotes cardiac hypertrophy by activating the autophagic degradation of SERCA2a. Commun Biol, 2025, 8(1): 596.
pmid: 40210720 |
| [94] |
Nian FL, Chen YY, Xia QY, Zhu C, Wu LY, Lu XL. Gut microbiota metabolite trimethylamine N-oxide promoted NAFLD progression by exacerbating intestinal barrier disruption and intrahepatic cellular imbalance. Int Immunopharmacol, 2024, 142(Pt B): 113173.
pmid: 39298816 |
| [95] |
Zhang Y, Zhang YJ, Jia JL, Peng HC, Qian Q, Pan ZL, Liu DY. Nitrite and nitrate in meat processing: functions and alternatives. Curr Res Food Sci, 2023, 6: 100470.
pmid: 36891544 |
| [96] |
Bouvard V, Loomis D, Guyton KZ, Grosse Y, Ghissassi FE, Benbrahim-Tallaa L, Guha N, Mattock H, Straif K, International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol, 2015, 16(16): 1599-1600.
pmid: 26514947 |
| [97] | Cai LF, Li N, Du S, Tan Y, Li K, Wang YL. Progress in hazards, synthesis and inhibition of N-nitroso compounds in vitro and in vivo. Food Sci, 2016, 37(5): 271-277. |
| 蔡鲁峰, 李娜, 杜莎, 谭雅, 李珂, 王远亮. N-亚硝基化合物的危害及其在体内外合成和抑制的研究进展. 食品科学, 2016, 37(5): 271-277. | |
| [98] |
Li YP, Hecht SS. Metabolic activation and DNA interactions of carcinogenic N-nitrosamines to which humans are commonly exposed. Int J Mol Sci, 2022, 23(9): 4559.
pmid: 35562949 |
| [99] |
Yamazaki H, Oda Y, Funae Y, Imaoka S, Inui Y, Guengerich FP, Shimada T. Participation of rat liver cytochrome P450 2E1 in the activation of N-nitrosodimethylamine and N-nitrosodiethylamine to products genotoxic in an acetyltransferase-overexpressing salmonella typhimurium strain (NM2009). Carcinogenesis, 1992, 13(6): 979-985.
pmid: 1600620 |
| [100] |
EFSA Panel on Contaminants in the Food Chain (EFSA CONTAM Panel), Schrenk D, Bignami M, Bodin L, Chipman JK, Del Mazo J, Hogstrand C, Hoogenboom LR, Leblanc JC, Nebbia CS, Nielsen E, Ntzani E, Petersen A, Sand S, Schwerdtle T, Vleminckx C, Wallace H, Romualdo B, Cristina F, Stephen H, Marco I, Mosbach-Schulz O, Riolo F, Christodoulidou A, Grasl-Kraupp B. Risk assessment of N-nitrosamines in food. EFSA J, 2023, 21(3): e07884.
pmid: 36999063 |
| [101] |
de la Monte SM, Neusner A, Chu J, Lawton M. Epidemiological trends strongly suggest exposures as etiologic agents in the pathogenesis of sporadic alzheimer’s disease, diabetes mellitus, and non-alcoholic steatohepatitis. J Alzheimers Dis, 2009, 17(3): 519-529.
pmid: 19363256 |
| [102] |
di Corcia M, Tartaglia N, Polito R, Ambrosi A, Messina G, Francavilla VC, Cincione RI, Della Malva A, Ciliberti MG, Sevi A, Messina G, Albenzio M. Functional properties of meat in athletes’ performance and recovery. Int J Environ Res Public Health, 2022, 19(9): 5145.
pmid: 35564540 |
| [103] | Domellöf M, Sjöberg A. Iron-a background article for the nordic nutrition recommendations 2023. Food Nutr Res, 2024, doi: 10.29219/fnr.v68.10451. |
| [104] |
Kalman D, Hewlings S, Madelyn-Adjei A, Ebersole B. Dietary heme iron: a review of efficacy, safety and tolerability. Nutrients, 2025, 17(13): 2132.
pmid: 40647237 |
| [105] |
Pantopoulos K. Oral iron supplementation: new formulations, old questions. Haematologica, 2024, 109(9): 2790-2801.
pmid: 38618666 |
| [106] |
Papier K, Fensom GK, Knuppel A, Appleby PN, Tong TYN, Schmidt JA, Travis RC, Key TJ, Perez-Cornago A. Meat consumption and risk of 25 common conditions: outcome-wide analyses in 475,000 men and women in the UK biobank study. BMC Med, 2021, 19(1): 53.
pmid: 33648505 |
| [107] |
Ruxton CHS, Gordon S. Animal board invited review: the contribution of red meat to adult nutrition and health beyond protein. Animal, 2024, 18(3): 101103.
pmid: 38442540 |
| [108] | Kumar R, Singh U, Tiwari A, Tiwari P, Sahu JK, Sharma S. Vitamin B12: strategies for enhanced production, fortified functional food products and health benefits. Process Biochem, 2023, 127: 44-55. |
| [109] |
Stefanache A, Lungu II, Butnariu IA, Calin G, Gutu C, Marcu C, Grierosu C, Goroftei ERB, Duceac LD, Dabija MG, Popa F, Damir D. Understanding how minerals contribute to optimal immune function. J Immunol Res, 2023, 2023: 3355733.
pmid: 37946846 |
| [110] |
Bano I, Hassan MF, Kieliszek M. A comprehensive review of selenium as a key regulator in thyroid health. Biol Trace Elem Res, 2025, doi: 10.1007/s12011-025-04653-7.
pmid: 40358914 |
| [111] |
Wang Y, Uffelman CN, Bergia RE, Clark CM, Reed JB, Cross TWL, Lindemann SR, Tang MH, Campbell WW. Meat consumption and gut microbiota: a scoping review of literature and systematic review of randomized controlled trials in adults. Adv Nutr, 2023, 14(2): 215-237.
pmid: 36822879 |
| [112] |
O’Connor LE, Kim JE, Campbell WW. Total red meat intake of ≥0.5 servings/d does not negatively influence cardiovascular disease risk factors: a systemically searched meta-analysis of randomized controlled trials. Am J Clin Nutr, 2017, 105(1): 57-69.
pmid: 27881394 |
| [113] | Yu HC, Zhang JJ, Xie JC, Li XL, Wang YJ, Yang YJ, Zhou FS, Liu MC, Zhang Z, Shan ZL, Liu G, Pan A. Dose-response meta-analysis on risk of diabetes in relation to red and processed meat consumption—Asian populations, 2006-2021. China CDC Wkly, 2023, 5(45): 1012-1016. |
| [114] |
Ye KX, Cao C, Lin X, O’Brien KO, Gu ZL. Natural selection on HFE in asian populations contributes to enhanced non-heme iron absorption. BMC Genet, 2015, 16: 61.
pmid: 26054392 |
| [115] |
Fleming JA, Kris-Etherton PM, Petersen KS, Baer DJ. Effect of varying quantities of lean beef as part of a Mediterranean-style dietary pattern on lipids and lipoproteins: a randomized crossover controlled feeding trial. Am J Clin Nutr, 2021, 113(5): 1126-1136.
pmid: 33826691 |
| [116] |
O’Connor LE, Paddon-Jones D, Wright AJ, Campbell WW. A Mediterranean-style eating pattern with lean, unprocessed red meat has cardiometabolic benefits for adults who are overweight or obese in a randomized, crossover, controlled feeding trial. Am J Clin Nutr, 2018, 108(1): 33-40.
pmid: 29901710 |
| [117] |
Jomova K, Alomar SY, Alwasel SH, Nepovimova E, Kuca K, Valko M. Several lines of antioxidant defense against oxidative stress: antioxidant enzymes, nanomaterials with multiple enzyme-mimicking activities, and low-molecular-weight antioxidants. Arch Toxicol, 2024, 98(5): 1323-1367.
pmid: 38483584 |
| [118] |
Zaramela LS, Martino C, Alisson-Silva F, Rees SD, Diaz SL, Chuzel L, Ganatra MB, Taron CH, Secrest P, Zuñiga C, Huang JB, Siegel D, Chang G, Varki A, Zengler K. Gut bacteria responding to dietary change encode sialidases that exhibit preference for red meat-associated carbohydrates. Nat Microbiol, 2019, 4(12): 2082-2089.
pmid: 31548686 |
| [119] |
Lin XS, Yao HL, Guo JY, Huang YY, Wang WJ, Yin BR, Li XA, Wang T, Li CB, Xu XL, Zhou GH, Voglmeir J, Liu L. Protein glycosylation and gut microbiota utilization can limit the in vitro and in vivo metabolic cellular incorporation of Neu5Gc. Mol Nutr Food Res, 2022, 66(5): e2100615.
pmid: 34921741 |
| [120] |
Cantero MA, Guedes MRA, Fernandes R, Lollo PCB. Trimethylamine N-oxide reduction is related to probiotic strain specificity: a systematic review. Nutr Res, 2022, 104: 29-35.
pmid: 35588611 |
| [121] |
Wang QQ, Guo M, Liu Y, Xu MS, Shi LT, Li X, Zhao JX, Zhang H, Wang G, Chen W. Bifidobacterium breve and bifidobacterium longum attenuate choline-induced plasma trimethylamine N-oxide production by modulating gut microbiota in mice. Nutrients, 2022, 14(6): 1222.
pmid: 35334879 |
| [122] | Ahmad Kamal NH, Selamat J, Sanny M. Simultaneous formation of polycyclic aromatic hydrocarbons (PAHs) and heterocyclic aromatic amines (HCAs) in gas- grilled beef satay at different temperatures. Food Addit Contam: Part A, 2018, 35(5): 848-869. |
| [123] | Adeyeye SAO, Ashaolu TJ. Polycyclic aromatic hydrocarbons formation and mitigation in meat and meat products. Polycyclic Aromat Compd, 2022, 42(6): 3401-3411. |
| [124] | Salihi KAA, AL-Mhsenawi ZKF, Awad ZH, Ali RM. A comparative study between red and white meat: a literature review. Mirror Res Vet Sci Anim, 2023, 12(2): 1-9. |
| [125] |
Seiwert N, Heylmann D, Hasselwander S, Fahrer J. Mechanism of colorectal carcinogenesis triggered by heme iron from red meat. Biochim Biophys Acta Rev Cancer, 2020, 1873(1): 188334.
pmid: 31783067 |
| [126] |
Perota A, Galli C. N-glycolylneuraminic acid (Neu5Gc) null large animals by targeting the CMP-Neu5Gc hydroxylase (CMAH). Front Immunol, 2019, 10: 2396.
pmid: 31681287 |
| [127] |
Peri S, Kulkarni A, Feyertag F, Berninsone PM, Alvarez-Ponce D. Phylogenetic distribution of CMP- Neu5Ac hydroxylase (CMAH), the enzyme synthetizing the proinflammatory human xenoantigen Neu5Gc. Genome Biol Evol, 2018, 10(1): 207-219.
pmid: 29206915 |
| [128] |
Warrier M, Shih DM, Burrows AC, Ferguson D, Gromovsky AD, Brown AL, Marshall S, McDaniel A, Schugar RC, Wang ZN, Sacks J, Rong X, de Aguiar Vallim T, Chou J, Ivanova PT, Myers DS, Brown HA, Lee RG, Crooke RM, Graham MJ, Liu XL, Parini P, Tontonoz P, Lusis AJ, Hazen SL, Temel RE, Brown JM. The TMAO-generating enzyme flavin monooxygenase 3 is a central regulator of cholesterol balance. Cell Rep, 2015, 10(3): 326-338.
pmid: 25600868 |
| [129] |
Shan ZL, Sun TP, Huang H, Chen SJ, Chen LK, Luo C, Yang W, Yang XF, Yao P, Cheng JQ, Hu FB, Liu LG. Association between microbiota-dependent metabolite trimethylamine-N-oxide and type 2 diabetes. Am J Clin Nutr, 2017, 106(3): 888-894.
pmid: 28724646 |
| [130] | Tang WHW, Wang ZN, Li XMS, Fan YY, Li DS, Wu YP, Hazen SL. Increased trimethylamine N-oxide portends high mortality risk independent of glycemic control in patients with type 2 diabetes mellitus. Clin Chem, 2017, 63(1): 297-306. |
| [131] |
Ye ZX, Chen LL, Zeng XC, Fang Q, Zheng BJ, Luo CY, Rao T, Ouyang DS. TMAO as a potential biomarker and therapeutic target for chronic kidney disease: a review. Front Pharmacol, 2022, 13: 929262.
pmid: 36034781 |
| [132] |
Zhang WC, Miikeda A, Zuckerman J, Jia X, Charugundla S, Zhou ZQ, Kaczor-Urbanowicz KE, Magyar C, Guo FF, Wang ZN, Pellegrini M, Hazen SL, Nicholas SB, Lusis AJ, Shih DM. Inhibition of microbiota-dependent TMAO production attenuates chronic kidney disease in mice. Sci Rep, 2021, 11(1): 518.
pmid: 33436815 |
| [133] |
Theofilis P, Vordoni A, Kalaitzidis RG. Trimethylamine N-oxide levels in non-alcoholic fatty liver disease: a systematic review and meta-analysis. Metabolites, 2022, 12(12): 1243.
pmid: 36557281 |
| [134] |
Dai X, Hou HQ, Zhang WR, Liu TY, Li Y, Wang SN, Wang BM, Cao HL. Microbial metabolites: critical regulators in NAFLD. Front Microbiol, 2020, 11: 567654.
pmid: 33117316 |
| [135] |
Lombardo M, Aulisa G, Marcon D, Rizzo G, Tarsisano MG, Di Renzo L, Federici M, Caprio M, De Lorenzo A. Association of urinary and plasma levels of trimethylamine N-oxide (TMAO) with foods. Nutrients, 2021, 13(5): 1426.
pmid: 33922680 |
| [136] | Tomczyk M, Heileson JL, Babiarz M, Calder PC. Athletes can benefit from increased intake of EPA and DHA—evaluating the evidence. Nutrients, 2023, 15(23): 4925. |
| [137] |
Pisaniello AD, Psaltis PJ, King PM, Liu G, Gibson RA, Tan JT, Duong M, Nguyen T, Bursill CA, Worthley MI, Nicholls SJ, Di Bartolo BA. Omega-3 fatty acids ameliorate vascular inflammation: a rationale for their atheroprotective effects. Atherosclerosis, 2021, 324: 27-37.
pmid: 33812168 |
| [138] |
Allam-Ndoul B, Guénard F, Barbier O, Vohl MC. Effect of n-3 fatty acids on the expression of inflammatory genes in THP-1 macrophages. Lipids Health Dis, 2016, 15(1): 69.
pmid: 27044314 |
| [139] |
Zhang TT, Xu J, Wang YM, Xue CH. Health benefits of dietary marine DHA/EPA-enriched glycerophospholipids. Prog Lipid Res, 2019, 75: 100997.
pmid: 31442526 |
| [140] |
Xu ZJ, Li Q, Ding L, Shi HH, Xue CH, Mao XZ, Wang YM, Zhang TT. A comparative study of the effects of phosphatidylserine rich in DHA and EPA on Aβ-induced Alzheimer’s disease using cell models. Food Funct, 2021, 12(10): 4411-4423.
pmid: 33876786 |
| [141] |
Wu ZJ, Li YC, Zheng Y, Zhou MQ, Li H, Wu SX, Zhao XY, Yang YH, Du L. Differential effects of EPA and DHA on aging-related sarcopenia in mice and possible mechanisms involved. Food Funct, 2025, 16(2): 601-616.
pmid: 39704327 |
| [142] |
Wei WT, Hu MJ, Huang J, Yu SY, Li XD, Li YH, Mao LM. Anti-obesity effects of DHA and EPA in high fat-induced insulin resistant mice. Food Funct, 2021, 12(4): 1614-1625.
pmid: 33476356 |
| [143] |
Zhu J, Song SX, Xu XL, Zhou GH, Li CB. White meat proteins were more conducive to hepatic antioxidative status than soybean and red meat proteins. J Food Biochem, 2022, 46(4): e13947.
pmid: 34561892 |
| [144] |
Maky MA, Zendo T. Identification of a novel bioactive peptide derived from frozen chicken breast hydrolysate and the utilization of hydrolysates as biopreservatives. Biology (Basel), 2023, 12(9): 1218.
pmid: 37759617 |
| [145] | Clinton SK, Giovannucci EL, Hursting SD. The World Cancer Research Fund/American Institute for Cancer Research third expert report on diet, nutrition, physical activity, and cancer: impact and future directions. J Nutr, 2020, 150(4): 663-671. |
| [146] |
Liu WX, Luo XL, Huang Y, Zhao MJ, Liu T, Wang J, Feng FQ. Influence of cooking techniques on food quality, digestibility, and health risks regarding lipid oxidation. Food Res Int, 2023, 167: 112685.
pmid: 37087258 |
| [147] | Pathare PB, Roskilly AP. Quality and energy evaluation in meat cooking. Food Eng Rev, 2016, 8(4): 435-447. |
| [148] |
Chen S, Jiang PP, Yu DX, Liao GC, Wu SL, Fang AP, Chen PY, Wang XY, Luo Y, Long JA, Zhong RH, Liu ZY, Li CL, Zhang DM, Zhu HL. Effects of probiotic supplementation on serum trimethylamine-N-oxide level and gut microbiota composition in young males: a double-blinded randomized controlled trial. Eur J Nutr, 2021, 60(2): 747-758.
pmid: 32440731 |
| [149] |
Liang X, Zhang Z, Lv YY, Tong LJ, Liu TJ, Yi HX, Zhou XH, Yu Z, Tian XY, Cui QY, Yang JB, Gong PM, Zhang LW. Reduction of intestinal trimethylamine by probiotics ameliorated lipid metabolic disorders associated with atherosclerosis. Nutrition, 2020, 79-80: 110941.
pmid: 32858376 |
| [150] |
Ramireddy L, Tsen HY, Chiang YC, Hung CY, Wu SR, Young SL, Lin JS, Huang CH, Chiu SH, Chen CC, Chen CC. Molecular identification and selection of probiotic strains able to reduce the serum TMAO level in mice challenged with choline. Foods, 2021, 10(12): 2931.
pmid: 34945482 |
| [151] |
Abebe BK, Guo JT, Jilo DD, Wang JF, Yu SC, Liu HB, Cheng G, Zan LS. Transforming beef quality through healthy breeding: a strategy to reduce carcinogenic compounds and enhance human health: a review. Mamm Genome, 2025, 36(3): 787-811.
pmid: 40343484 |
| [152] |
Mahfuz S, Mun HS, Dilawar MA, Ampode KMB, Yang CJ. Potential role of protocatechuic acid as natural feed additives in farm animal production. Animals (Basel), 2022, 12(6): 741.
pmid: 35327138 |
| [153] |
Serra V, Salvatori G, Pastorelli G. Dietary polyphenol supplementation in food producing animals: effects on the quality of derived products. Animals (Basel), 2021, 11(2): 401.
pmid: 33562524 |
| [1] | 肖诚, 刘洁颖, 杨春如, 于淼. LMNA基因突变相关脂肪萎缩综合征的研究进展[J]. 遗传, 2022, 44(10): 913-925. |
| [2] | 刘林玉, 杜司晨, 张进, 马端. 戈谢氏病致病机制及治疗方法[J]. 遗传, 2015, 37(6): 510-516. |
| [3] | 许烨, 张嘉莹, 杨博宇, 何志宏, 张慕晨, 于珍, 顾鸣敏. 腓骨肌萎缩症4型遗传学研究进展[J]. 遗传, 2015, 37(6): 501-509. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||

www.chinagene.cn
备案号:京ICP备09063187号-4
总访问:,今日访问:,当前在线:
