Hereditas(Beijing) ›› 2020, Vol. 42 ›› Issue (10): 1017-1027.doi: 10.16288/j.yczz.20-138
• Research Article • Previous Articles Next Articles
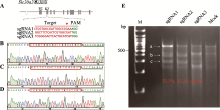
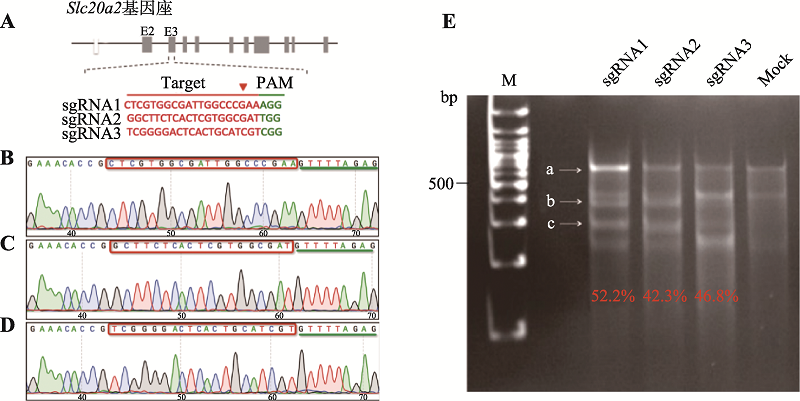
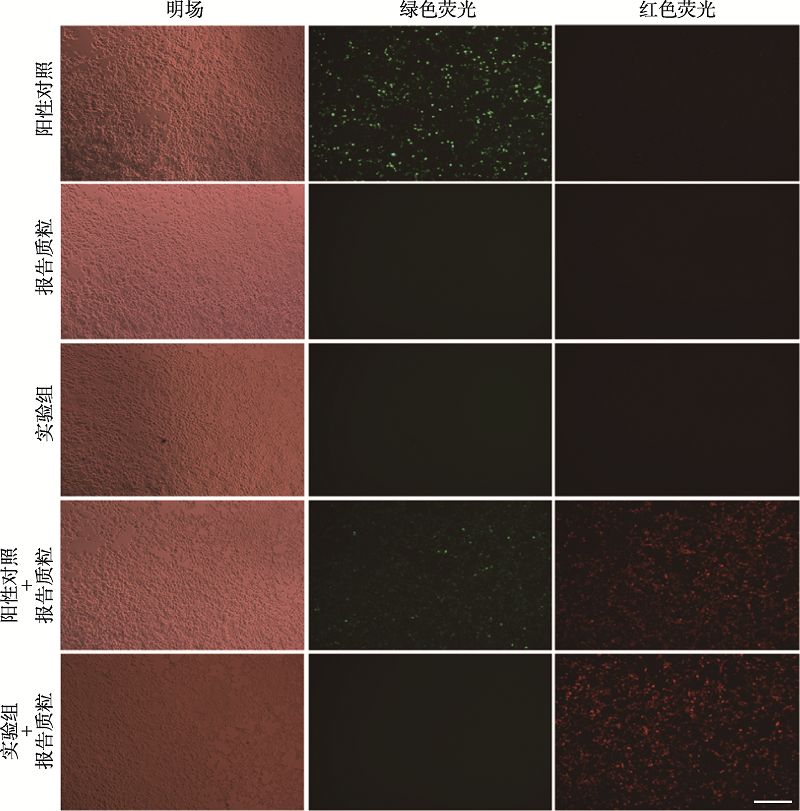
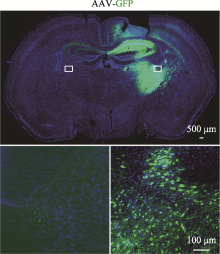
Construction of a striatum-specific Slc20a2 gene knockout mice model by CRISPR/Cas9 AAV system
Minting Lin1,2, Lulu Lai1, Miao Zhao1, Biwei Lin1, Xiangping Yao1,3( )
)
- 1. Department of Neurology, the First Affiliated Hospital of Fujian Medical University, Fuzhou 350005, China
2. Institute of Neurosciences, Fujian Medical University, Fuzhou 350004, China
3. Fujian Key Laboratory of Molecular Neurology, Fuzhou 350005, China
-
Received:2020-05-18Revised:2020-08-18Online:2020-10-20Published:2020-10-12 -
Contact:Yao Xiangping E-mail:119373522@qq.com -
Supported by:Supported by the National Natural Science Foundation of China No)(81801129);Startup Fund for Scientific Research of Fujian Medical University Nos(2017XQ1071);Startup Fund for Scientific Research of Fujian Medical University Nos(2018QH2035)
Cite this article
Minting Lin, Lulu Lai, Miao Zhao, Biwei Lin, Xiangping Yao. Construction of a striatum-specific Slc20a2 gene knockout mice model by CRISPR/Cas9 AAV system[J]. Hereditas(Beijing), 2020, 42(10): 1017-1027.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] | Nicolas G, Pottier C, Charbonnier C, Guyant-Marechal L, Le Ber I, Pariente J, Labauge P, Ayrignac X, Defebvre L, Maltete D, Martinaud O, Lefaucheur R, Guillin O, Wallon D, Chaumette B, Rondepierre P, Derache N, Fromager G, Schaeffer S, Krystkowiak P, Verny C, Jurici S, Sauvee M, Verin M, Lebouvier T, Rouaud O, Thauvin-Robinet C, Rousseau S, Rovelet-Lecrux A, Frebourg T, Campion D, Hannequin D . Phenotypic spectrum of probable and genetically-confirmed idiopathic basal ganglia calcification. Brain, 2013,1369(11):3395-3407. |
| [2] |
Oliveira JRM, Spiteri E, Sobrido MJ, Hopfer S, Klepper J, Voit T, Gilbert J, Wszolek ZK, Calne DB, Stoessl AJ, Hutton M, Manyam BV, Boller F, Baquero M, Geschwind DH . Genetic heterogeneity in familial idiopathic basal ganglia calcification (Fahr disease). Neurology, 2004,63(11):2165-2167.
doi: 10.1212/01.wnl.0000145601.88274.88 pmid: 15596772 |
| [3] |
Wang C, Li YL, Shi L, Ren J, Patti M, Wang T, de Oliveira JRm, Sobrido MJ, Quintáns B, Baquero M, Cui XN, Zhang XY, Wang LQ, Xu HB, Wang JH, Yao J, Dai XH, Liu J, Zhang L, Ma HY, Gao Y, Ma XX, Feng SL, Liu MG, Wang QK, Forster IC, Zhang X, Liu JY. Mutations in SLC20A2 link familial idiopathic basal ganglia calcification with phosphate homeostasis. Nat Genet, 2012,44(3):254-256.
pmid: 22327515 |
| [4] |
Nicolas G, Pottier C, Maltete D, Coutant S, Rovelet- Lecrux A, Legallic S, Rousseau S, Vaschalde Y, Guyant- Marechal L, Augustin J, Martinaud O, Defebvre L, Krystkowiak P, Pariente J, Clanet M, Labauge P, Ayrignac X, Lefaucheur R, Le Ber I, Frébourg T, Hannequin D, Campion D . Mutation of the PDGFRB gene as a cause of idiopathic basal ganglia calcification. Neurology, 2013,80(2):181-187.
doi: 10.1212/WNL.0b013e31827ccf34 |
| [5] |
Keller A, Westenberger A, Sobrido MJ, Garcia-Murias M, Domingo A, Sears RL, Lemos RR, Ordoñez-Ugalde A, Nicolas G, da Cunha JE, Rushing EJ, Hugelshofer M, Wurnig MC, Kaech A, Reimann R, Lohmann K, Dobričić V, Carracedo A, Petrović I, Miyasaki JM, Abakumova I, Mäe MA, Raschperger E, Zatz M, Zschiedrich K, Klepper J, Spiteri E, Prieto JM, Navas I, Preuss M, Dering C, Janković M, Paucar M, Svenningsson P, Saliminejad K, Khorshid HR, Novaković I, Aguzzi A, Boss A, Le Ber I, Defer G, Hannequin D, Kostić VS, Campion D, Geschwind DH, Coppola G, Betsholtz C, Klein C, Oliveira JR. Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. Nat Genet, 2013,45(9):1077-1082.
doi: 10.1038/ng.2723 pmid: 23913003 |
| [6] |
Legati A, Giovannini D, Nicolas G, López-Sánchez U, Quintáns B, Oliveira JR, Sears RL, Ramos EM, Spiteri E, Sobrido MJ, Carracedo A, Castro-Fernández C, Cubizolle S, Fogel BL, Goizet C, Jen JC, Kirdlarp S, Lang AE, Miedzybrodzka Z, Mitarnun W, Paucar M, Paulson H, Pariente J, Richard AC, Salins NS, Simpson SA, Striano P, Svenningsson P, Tison F, Unni VK, Vanakker O, Wessels MW, Wetchaphanphesat S, Yang M, Boller F, Campion D, Hannequin D, Sitbon M, Geschwind DH, Battini JL, Coppola G . Mutations in XPR1 cause primary familial brain calcification associated with altered phosphate export. Nat Genet, 2015,47(6):579-581.
doi: 10.1038/ng.3289 pmid: 25938945 |
| [7] |
Yao XP, Cheng XW, Wang C, Zhao M, Guo XX, Su HZ, Lai LL, Zou XH, Chen XJ, Zhao YY, Dong EL, Lu YQ, Wu S, Li XJ, Fan GF, Yu HJ, Xu JF, Wang N, Xiong ZQ, Chen WJ. Biallelic mutations in MYORG cause autosomal recessive primary familial brain calcification. Neuron, 2018, 98(6): 1116-1123.e5.
doi: 10.1016/j.neuron.2018.05.037 pmid: 29910000 |
| [8] |
Cen ZD, Chen Y, Chen S, Wang H, Yang DH, Zhang HM, Wu HW, Wang LB, Tang SY, Ye J, Shen J, Wang HT, Fu F, Chen XH, Xie F, Liu P, Xu X, Cao JZ, Cai P, Pan QQ, Li JY, Yang W, Shan PF, Li YZ, Liu JY, Zhang BR, Luo W . Biallelic loss-of-function mutations in JAM2 cause primary familial brain calcification. Brain, 2020,143(2):491-502.
doi: 10.1093/brain/awz392 pmid: 31851307 |
| [9] |
Lemos RR, Ramos EM, Legati A, Nicolas G, Jenkinson EM, Livingston JH, Crow YJ, Campion D, Coppola G, Oliveira JRM . Update and mutational analysis of SLC20A2: a major cause of primary familial brain calcification. Hum Mutat, 2015,36(5):489-495.
doi: 10.1002/humu.22778 pmid: 25726928 |
| [10] |
Yamada M, Tanaka M, Takagi M, Kobayashi S, Taguchi Y, Takashima S, Tanaka K, Touge T, Hatsuta H, Murayama S, Hayashi Y, Kaneko M, Ishiura H, Mitsui J, Atsuta N, Sobue G, Shimozawa N, Inuzuka T, Tsuji S, Hozumi I . Evaluation of SLC20A2 mutations that cause idiopathic basal ganglia calcification in Japan. Neurology, 2014,82(8):705-712.
doi: 10.1212/WNL.0000000000000143 |
| [11] |
Jensen N, Schrøder HD, Hejbøl EK, Füchtbauer EM, de Oliveira JR, Pedersen L. Loss of function of Slc20a2 associated with familial idiopathic basal ganglia calcification in humans causes brain calcifications in mice. J Mol Neurosci, 2013,51(3):994-999.
doi: 10.1007/s12031-013-0085-6 |
| [12] |
Wallingford MC, Gammill HS, Giachelli CM . Slc20a2 deficiency results in fetal growth restriction and placental calcification associated with thickened basement membranes and novel CD13 and lamininα1 expressing cells. Reprod Biol, 2016,16(1):13-26.
doi: 10.1016/j.repbio.2015.12.004 pmid: 26952749 |
| [13] |
Platt RJ, Chen SD, Zhou Y, Yim MJ, Swiech L, Kempton HR, Dahlman JE, Parnas O, Eisenhaure TM, Jovanovic M, Graham DB, Jhunjhunwala S, Heidenreich M, Xavier RJ, Langer R, Anderson DG, Hacohen N, Regev A, Feng GP, Sharp PA, Zhang F . CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell, 2014,159(2):440-455.
doi: 10.1016/j.cell.2014.09.014 |
| [14] |
Wallingford MC, Chia JJ, Leaf EM, Borgeia S, Chavkin NW, Sawangmake C, Marro K, Cox TC, Speer MY, Giachelli CM . SLC20A2 deficiency in mice leads to elevated phosphate levels in cerbrospinal fluid and glymphatic pathway-associated arteriolar calcification, and recapitulates human idiopathic basal ganglia calcification. Brain Pathol, 2017,27(1):64-76.
doi: 10.1111/bpa.12362 pmid: 26822507 |
| [15] |
Bezerra DP, Oliveira JRM . New studies on knockout mouse for the SLC20A2 gene show much more than brain calcifications. J Mol Neurosci, 2016,59(4):565-566.
doi: 10.1007/s12031-016-0778-8 pmid: 27380911 |
| [16] |
Forster I, Hernando N, Sorribas V, Werner A . Phosphate transporters in renal, gastrointestinal, and other tissues. Adv Chronic Kidney Dis, 2011,18(2):63-76.
doi: 10.1053/j.ackd.2011.01.006 pmid: 21406290 |
| [17] |
Villa-Bellosta R, Ravera S, Sorribas V, Stange G, Levi M, Murer H, Biber J, Forster IC . The Na+-Pi cotransporter PiT-2 ( SLC20A2) is expressed in the apical membrane of rat renal proximal tubules and regulated by dietary Pi. Am J Physiol Renal Physiol, 2009,296(4):F691-699.
doi: 10.1152/ajprenal.90623.2008 pmid: 19073637 |
| [18] |
Villa-Bellosta R, Sorribas V . Compensatory regulation of the sodium/phosphate cotransporters NaPi-IIc ( SCL34A3) and Pit-2(SLC20A2) during Pi deprivation and acidosis. Pflugers Arch, 2010,459(3):499-508.
doi: 10.1007/s00424-009-0746-z pmid: 19841935 |
| [19] | Wang JP, Zhang YM . The application of Red/ET recombination to high efficient gene-targeting vector construction. Hereditas (Beijing), 2005,27(6):953-958. |
| 王军平, 张友明 . Red/ET重组在基因打靶载体快速构建中的应用. 遗传, 2005,27(6):953-958. | |
| [20] | He XB, Gu F . Genome-editing: focus on the off-target effects. Chin J Biotechnol, 2017,33(10):1757-1775. |
| 何秀斌, 谷峰 . 基因组编辑脱靶研究进展. 生物工程学报, 2017,33(10):1757-1775. | |
| [21] |
Jensen N, Schrøder HD, Hejbøl EK, Thomsen JS, Brüel A, Larsen FT, Vinding MC, Orlowski D, Füchtbauer EM, Oliveira JRM, Pedersen L . Mice knocked out for the primary brain calcification-associated gene Slc20a2 show unimpaired prenatal survival but retarded growth and nodules in the brain that grow and calcify over time. Am J Pathol, 2018,188(8):1865-1881.
doi: 10.1016/j.ajpath.2018.04.010 pmid: 29803831 |
| [1] | Bingzheng Wang, Chao Zhang, Jiali Zhang, Jin Sun. Conditional editing of the Drosophila melanogaster genome using single transcripts expressing Cas9 and sgRNA [J]. Hereditas(Beijing), 2023, 45(7): 593-601. |
| [2] | Lan Wang, Fan Zeng, Rongfeng Huang, Shu Lin, Zhihui Zhang, Min-Dian Li. Adipocyte reconstitution of Npy4r gene in Npy4r silenced mice promotes diet-induced obesity [J]. Hereditas(Beijing), 2023, 45(2): 144-155. |
| [3] | Meizhen Liu, Liren Wang, Yongmei Li, Xueyun Ma, Honghui Han, Dali Li. Generation of genetically modified rat models via the CRISPR/Cas9 technology [J]. Hereditas(Beijing), 2023, 45(1): 78-87. |
| [4] | Xiaojun Zhang, Kun Xu, Juncen Shen, Lu Mu, Hongrun Qian, Jieyu Cui, Baoxia Ma, Zhilong Chen, Zhiying Zhang, Zehui Wei. A CRISPR/Cas9-Gal4BD donor adapting system for enhancing homology-directed repair [J]. Hereditas(Beijing), 2022, 44(8): 708-719. |
| [5] | Chong Zhang, Zixuan Wei, Min Wang, Yaosheng Chen, Zuyong He. Editing MC1R in human melanoma cells by CRISPR/Cas9 and functional analysis [J]. Hereditas(Beijing), 2022, 44(7): 581-590. |
| [6] | Yao Liu, Xianhui Zhou, Shuhong Huang, Xiaolong Wang. Prime editing: a search and replace tool with versatile base changes [J]. Hereditas(Beijing), 2022, 44(11): 993-1008. |
| [7] | Yuting Han, Bowen Xu, Yutong Li, Xinyi Lu, Xizhi Dong, Yuhao Qiu, Qinyun Che, Ruibao Zhu, Li Zheng, Xiaochen Li, Xu Si, Jianquan Ni. The cutting edge of gene regulation approaches in model organism Drosophila [J]. Hereditas(Beijing), 2022, 44(1): 3-14. |
| [8] | Guangwu Yang, Yuan Tian. The F-box gene Ppa promotes lipid storage in Drosophila [J]. Hereditas(Beijing), 2021, 43(6): 615-622. |
| [9] | Dingwei Peng, Ruiqiang Li, Wu Zeng, Min Wang, Xuan Shi, Jianhua Zeng, Xiaohong Liu, Yaoshen Chen, Zuyong He. Editing the cystine knot motif of MSTN enhances muscle development of Liang Guang Small Spotted pigs [J]. Hereditas(Beijing), 2021, 43(3): 261-270. |
| [10] | Na Wang, Zhilian Jia, Qiang Wu. RFX5 regulates gene expression of the Pcdhα cluster [J]. Hereditas(Beijing), 2020, 42(8): 760-774. |
| [11] | Guoling Li, Shanxin Yang, Zhenfang Wu, Xianwei Zhang. Recent developments in enhancing the efficiency of CRISPR/Cas9- mediated knock-in in animals [J]. Hereditas(Beijing), 2020, 42(7): 641-656. |
| [12] | Yingnan Chen, Jing Lu. Application of CRISPR/Cas9 mediated gene editing in trees [J]. Hereditas(Beijing), 2020, 42(7): 657-668. |
| [13] | Siyuan Liu, Guoqiang Yi, Zhonglin Tang, Bin Chen. Progress on genome-wide CRISPR/Cas9 screening for functional genes and regulatory elements [J]. Hereditas(Beijing), 2020, 42(5): 435-443. |
| [14] | Liwen Bao, Yiye Zhou, Fanyi Zeng. Advances in gene therapy for β-thalassemia and hemophilia based on the CRISPR/Cas9 technology [J]. Hereditas(Beijing), 2020, 42(10): 949-964. |
| [15] | Peifeng Liu, Qiang Wu. Probing 3D genome by CRISPR/Cas9 [J]. Hereditas(Beijing), 2020, 42(1): 18-31. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||